| |||
| Names | |||
|---|---|---|---|
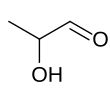
| IUPAC name 2-Hydroxypropanal | |||
| Other names Hydroxypropionaldehyde | |||
| Identifiers | |||
| CAS Number | |||
| 3D model (JSmol) | |||
| ChEBI | |||
| ChemSpider | |||
| ECHA InfoCard | 100.237.284 | ||
| KEGG | |||
| PubChem CID | |||
| UNII | |||
| CompTox Dashboard (EPA) | |||
InChI
| |||
SMILES
| |||
| Properties | |||
| Chemical formula | C3H6O2 | ||
| Molar mass | 74.079 g·mol | ||
| Related compounds | |||
| Related aldehydes | Glycolaldehyde | ||
| Except where otherwise noted, data are given for materials in their standard state (at 25 °C , 100 kPa).
| |||
Lactaldehyde is an intermediate in the methylglyoxal metabolic pathway. Methylglyoxal is converted to D-lactaldehyde by glycerol dehydrogenase (gldA). Lactaldehyde is then oxidized to lactic acid by aldehyde dehydrogenase.
Structure
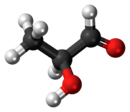
Lactaldehyde is a three-carbon atom species with a carbonyl group on the first carbon atom (making it an aldehyde), and a hydroxy group on the second carbon atom, making it a secondary alcohol. The molecule is chiral, its stereocenter being located on the second carbon atom.
Lactaldehyde exists in several forms: in open-chain form and as cyclic hemiacetal; in solution and in crystal forms; as monomer and as dimer. In crystal form, three conformers occur as hemiacetal dimers with a 1,4-dioxane ring skeleton:

In equilibrium solution, negligibly small amounts of the monomer and at least one five-membered ring dimer exist.
References
- Huang PC; Miller ON (1958). "The metabolism of lactaldehyde, page 205" (PDF). J. Biol. Chem. 231 (1): 201–5. doi:10.1016/S0021-9258(19)77298-6. PMID 13538961.
- Takahashi, H (1983). "Conformational studies of DL-lactaldehyde by H-NMR, Raman and i.r. spectroscopy". Spectrochimica Acta Part A: Molecular Spectroscopy. 39 (6): 569–572. doi:10.1016/0584-8539(83)80108-1.