| Revision as of 21:19, 18 June 2011 editRjwilmsi (talk | contribs)Extended confirmed users, Pending changes reviewers, Rollbackers932,067 edits doi:10.1016/S0045-6535(98)00041-1← Previous edit | Latest revision as of 08:07, 25 March 2023 edit undoCitation bot (talk | contribs)Bots5,427,483 edits Alter: title. Add: pmid, page, issue, bibcode. | Use this bot. Report bugs. | Suggested by Abductive | #UCB_webform 2854/3850 | ||
| (29 intermediate revisions by 24 users not shown) | |||
| Line 1: | Line 1: | ||
| {{chembox | {{chembox | ||
| | Verifiedfields = changed | |||
| ⚫ | | verifiedrevid = |
||
| | Watchedfields = changed | |||
| ⚫ | | |
||
| ⚫ | | verifiedrevid = 434992161 | ||
| ⚫ | | |
||
| ⚫ | | ImageFile = Retene.svg | ||
| ⚫ | | |
||
| ⚫ | | ImageSize = | ||
| ⚫ | | |
||
| ⚫ | | ImageName = Skeletal formula | ||
| ⚫ | | |
||
| ⚫ | | ImageFile1 = Retene-3D-balls.png | ||
| ⚫ | | |
||
| ⚫ | | ImageSize1 = 210px | ||
| | IUPACName = 7-Isopropyl-1-methylphenanthrene | |||
| ⚫ | | ImageName1 = Ball-and-stick model | ||
| ⚫ | | |
||
| | PIN = 1-Methyl-7-(propan-2-yl)phenanthrene | |||
| ⚫ | |Section1= |
||
| ⚫ | | OtherNames = Retene | ||
| ⚫ | | |
||
| ⚫ | |Section1={{Chembox Identifiers | ||
| | PubChem=10222 | |||
| | CASNo_Ref = {{cascite|correct|??}} | |||
| | SMILES=Cc3cccc2c1ccc (C(C)C)cc1ccc23 | |||
| ⚫ | | CASNo =483-65-8 | ||
| | UNII_Ref = {{fdacite|correct|FDA}} | |||
| | UNII = 0W2D2E1P9Q | |||
| | PubChem =10222 | |||
| | EINECS = 207-597-9 | |||
| | ChemSpiderID_Ref = {{chemspidercite|changed|chemspider}} | |||
| | ChemSpiderID = 9805 | |||
| | InChI = 1/C18H18/c1-12(2)14-7-10-17-15(11-14)8-9-16-13(3)5-4-6-18(16)17/h4-12H,1-3H3 | |||
| | InChIKey = NXLOLUFNDSBYTP-UHFFFAOYAL | |||
| | StdInChI_Ref = {{stdinchicite|changed|chemspider}} | |||
| | StdInChI = 1S/C18H18/c1-12(2)14-7-10-17-15(11-14)8-9-16-13(3)5-4-6-18(16)17/h4-12H,1-3H3 | |||
| | StdInChIKey_Ref = {{stdinchicite|changed|chemspider}} | |||
| | StdInChIKey = NXLOLUFNDSBYTP-UHFFFAOYSA-N | |||
| | SMILES =Cc1cccc3c1ccc2cc(C(C)C)ccc23 | |||
| }} | }} | ||
| |Section2= |
|Section2={{Chembox Properties | ||
| | |
| Formula =C<sub>18</sub>H<sub>18</sub> | ||
| | |
| MolarMass =234.33552 | ||
| | |
| Appearance = | ||
| | |
| Density = | ||
| | |
| MeltingPtC = 98.5 | ||
| | BoilingPtC = 390 | |||
| | BoilingPt=390 °C | |||
| | |
| Solubility = | ||
| ⚫ | |||
| ⚫ | |Section3= |
||
| | MainHazards= | |||
| | FlashPt= | |||
| | Autoignition= | |||
| }} | }} | ||
| ⚫ | |Section3={{Chembox Hazards | ||
| | MainHazards = | |||
| | FlashPt = | |||
| | AutoignitionPt = | |||
| ⚫ | }} | ||
| }} | }} | ||
| '''Retene''', methyl isopropyl phenanthrene or 1-methyl-7-isopropyl phenanthrene, C<sub>18</sub>H<sub>18</sub>, is a ] present in the ] fraction, ] above 360 °C. It occurs naturally in the ]s obtained by the ] of ]ous ]s. It crystallizes in large plates, which melt at 98.5 °C and boil at 390 °C. It is readily soluble in warm ] and in hot ]. ] and boiling ] reduce it to a |
'''Retene''', methyl isopropyl phenanthrene or 1-methyl-7-isopropyl phenanthrene, C<sub>18</sub>H<sub>18</sub>, is a ] present in the ] fraction, ] above 360 °C. It occurs naturally in the ]s obtained by the ] of ]ous ]s. It crystallizes in large plates, which melt at 98.5 °C and boil at 390 °C. It is readily soluble in warm ] and in hot ]. ] and boiling ] reduce it to a tetrahydroretene, but if it heated with ] and ] to 260 °C, a dodecahydride is formed. ] oxidizes it to retene quinone, ] and ]. It forms a ] that melts at 123-124 °C. | ||
| Retene is derived by degradation of specific ]s biologically produced by ] trees. The presence of traces of retene in the air is an indicator of ]s; it is a major product of ] of ] trees.<ref>{{cite web|last1=Simonich|first1=Staci|title=Community Outreach and Education Program Presents Unsolved Mysteries of Human Health: How Scientists Study Toxic Chemicals|url=http://www.unsolvedmysteries.oregonstate.edu/GCMS_04.shtml|publisher=Oregon State University|archive-url=https://web.archive.org/web/20080316183140/http://www.unsolvedmysteries.oregonstate.edu/GCMS_04.shtml|archive-date=16 March 2008|url-status=dead|language=en|quote=The presence of the chemical retene (REE-teen) in the air is a good indication of burning vegetation.}}</ref>{{Failed verification|date=September 2016|talk=Unsupported that a major part of conifer pyrolysis}} It is also present in ]s from ] and ]s.<ref>{{cite journal|journal=Chemosphere|volume=37|pages=219–235|year=1998|author=J. Koistinen, M. Lehtonena, K. Tukia, M. Soimasuo, M. Lahtiperab and A. Oikari|title=Identification of lipophilic pollutants discharged from a finnish pulp and paper mill |pmid=9650265 | doi=10.1016/S0045-6535(98)00041-1|issue=2|bibcode=1998Chmsp..37..219K }}</ref> | |||
| Retene is derived by degradation of specific ]s biologically produced by ] trees. | |||
| ⚫ | Retene, together with ], ] and ip-iHMN, is a ] of ]s, which makes it useful for ] analysis of rock ]s. The ratio of retene/cadalene in sediments can reveal the ratio of the genus '']'' in the biosphere.<ref>{{cite journal|journal=Geophysical Research Abstracts|volume=7|pages=10201|year=2005|title=Vascular plant biomarkers as ancient vegetation proxies and their stratigraphic use for tracing paleoclimatic changes during Jurassic in Western Europe|author=Y. Hautevelle, R. Michels, F. Malartre and A. Trouiller|url=http://www.cosis.net/abstracts/EGU05/10201/EGU05-J-10201.pdf|format=abstract}}</ref> | ||
| The presence of traces of retene in the air is an indicator of ]s; it is a major product of ] of ] trees.<ref>, Community Outreach and Education Program, Oregon State University</ref> It is also present in ]s from ] and ]s.<ref>{{cite journal|journal=Chemosphere|volume=37|pages=219–235|year=1998|title=IDENTIFICATION OF LIPOPHILIC POLLUTANTS DISCHARGED FROM A FINNISH PULP AND PAPER MILL|author=J. Koistinen, M. Lehtonena, K. Tukia, M. Soimasuo, M. Lahtiperab and A. Oikari|pmid=9650265 | doi=10.1016/S0045-6535(98)00041-1}}</ref> | |||
| ==Health effects== | |||
| ⚫ | Retene, together with ], ] and |
||
| A recent study has shown retene, which is a component of the Amazonian organic ], is cytotoxic to human lung cells.<ref>{{Cite journal|last=Nilmara de Oliveira Alves, Alexandre Teixeira Vessoni, Annabel Quinet, Rodrigo Soares Fortunato, Gustavo Satoru Kajitani, Milena Simões Peixoto, Sandra de Souza Hacon, Paulo Artaxo, Paulo Saldiva, Carlos Frederico Martins Menck, Silvia Regina Batistuzzo de Medeiros|date=September 7, 2017|title=Biomass burning in the Amazon region causes DNA damage and cell death in human lung cells.|journal=Scientific Reports|volume=7|issue=1 |page=10937 |doi=10.1038/s41598-017-11024-3|pmid=28883446 |pmc=5589902|bibcode=2017NatSR...710937D }}</ref> | |||
| ==References== | ==References== | ||
| <references/> | <references/> | ||
| {{1911|wstitle=Retene|volume=23|page=202}} | |||
| {{1911}} | |||
| ] | ] | ||
| ] | ] | ||
| ] | |||
| ] | |||
| ] | |||
Latest revision as of 08:07, 25 March 2023

| |
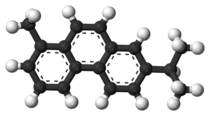
| |
| Names | |
|---|---|
| Preferred IUPAC name 1-Methyl-7-(propan-2-yl)phenanthrene | |
| Other names Retene | |
| Identifiers | |
| CAS Number | |
| 3D model (JSmol) | |
| ChemSpider | |
| ECHA InfoCard | 100.006.908 |
| EC Number |
|
| PubChem CID | |
| UNII | |
| CompTox Dashboard (EPA) | |
InChI
| |
SMILES
| |
| Properties | |
| Chemical formula | C18H18 |
| Molar mass | 234.33552 |
| Melting point | 98.5 °C (209.3 °F; 371.6 K) |
| Boiling point | 390 °C (734 °F; 663 K) |
| Except where otherwise noted, data are given for materials in their standard state (at 25 °C , 100 kPa).
| |
Retene, methyl isopropyl phenanthrene or 1-methyl-7-isopropyl phenanthrene, C18H18, is a polycyclic aromatic hydrocarbon present in the coal tar fraction, boiling above 360 °C. It occurs naturally in the tars obtained by the distillation of resinous woods. It crystallizes in large plates, which melt at 98.5 °C and boil at 390 °C. It is readily soluble in warm ether and in hot glacial acetic acid. Sodium and boiling amyl alcohol reduce it to a tetrahydroretene, but if it heated with phosphorus and hydriodic acid to 260 °C, a dodecahydride is formed. Chromic acid oxidizes it to retene quinone, phthalic acid and acetic acid. It forms a picrate that melts at 123-124 °C.
Retene is derived by degradation of specific diterpenoids biologically produced by conifer trees. The presence of traces of retene in the air is an indicator of forest fires; it is a major product of pyrolysis of conifer trees. It is also present in effluents from wood pulp and paper mills.
Retene, together with cadalene, simonellite and ip-iHMN, is a biomarker of vascular plants, which makes it useful for paleobotanic analysis of rock sediments. The ratio of retene/cadalene in sediments can reveal the ratio of the genus Pinaceae in the biosphere.
Health effects
A recent study has shown retene, which is a component of the Amazonian organic PM10, is cytotoxic to human lung cells.
References
- Simonich, Staci. "Community Outreach and Education Program Presents Unsolved Mysteries of Human Health: How Scientists Study Toxic Chemicals". Oregon State University. Archived from the original on 16 March 2008.
The presence of the chemical retene (REE-teen) in the air is a good indication of burning vegetation.
- J. Koistinen, M. Lehtonena, K. Tukia, M. Soimasuo, M. Lahtiperab and A. Oikari (1998). "Identification of lipophilic pollutants discharged from a finnish pulp and paper mill". Chemosphere. 37 (2): 219–235. Bibcode:1998Chmsp..37..219K. doi:10.1016/S0045-6535(98)00041-1. PMID 9650265.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - Y. Hautevelle, R. Michels, F. Malartre and A. Trouiller (2005). "Vascular plant biomarkers as ancient vegetation proxies and their stratigraphic use for tracing paleoclimatic changes during Jurassic in Western Europe" (abstract). Geophysical Research Abstracts. 7: 10201.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - Nilmara de Oliveira Alves, Alexandre Teixeira Vessoni, Annabel Quinet, Rodrigo Soares Fortunato, Gustavo Satoru Kajitani, Milena Simões Peixoto, Sandra de Souza Hacon, Paulo Artaxo, Paulo Saldiva, Carlos Frederico Martins Menck, Silvia Regina Batistuzzo de Medeiros (September 7, 2017). "Biomass burning in the Amazon region causes DNA damage and cell death in human lung cells". Scientific Reports. 7 (1): 10937. Bibcode:2017NatSR...710937D. doi:10.1038/s41598-017-11024-3. PMC 5589902. PMID 28883446.
{{cite journal}}: CS1 maint: multiple names: authors list (link)
![]() This article incorporates text from a publication now in the public domain: Chisholm, Hugh, ed. (1911). "Retene". Encyclopædia Britannica. Vol. 23 (11th ed.). Cambridge University Press. p. 202.
This article incorporates text from a publication now in the public domain: Chisholm, Hugh, ed. (1911). "Retene". Encyclopædia Britannica. Vol. 23 (11th ed.). Cambridge University Press. p. 202.