| Revision as of 06:59, 24 February 2013 editDavid Hedlund (talk | contribs)Extended confirmed users13,269 edits →Metabolism in humans← Previous edit | Latest revision as of 06:11, 30 December 2024 edit undoOAbot (talk | contribs)Bots441,761 editsm Open access bot: hdl updated in citation with #oabot. | ||
| (52 intermediate revisions by 38 users not shown) | |||
| Line 1: | Line 1: | ||
| {{Infobox |
{{Infobox medical condition (new) | ||
| | name = Toluene toxicity | |||
| | synonyms = | |||
| | image = Toluol.svg | |||
| | width = 80 | |||
| | caption = Chemical structure of ] | |||
| | pronounce = | |||
| | field = | |||
| | symptoms = | |||
| | complications = | |||
| | onset = | |||
| | duration = | |||
| ⚫ | |||
| | types = | |||
| | causes = | |||
| | risks = | |||
| | diagnosis = | |||
| | differential = | |||
| | prevention = | |||
| | treatment = | |||
| | medication = | |||
| | prognosis = | |||
| | frequency = | |||
| | deaths = | |||
| }} | }} | ||
| '''Toluene toxicity''' refers to the harmful effects caused by ] on the body. | '''Toluene toxicity''' refers to the harmful effects caused by ] on the body. | ||
| ==Metabolism in humans== | ==Metabolism in humans== | ||
| ⚫ | While a significant amount of ], 25%–40%, is exhaled unchanged via the lungs, a greater proportion is metabolised and excreted via other pathways. The primary route of toluene metabolism is by ] to ] by members of the ] (CYP) superfamily.<ref name=Shou>{{cite journal| last =Shou| first =M|author2=Lu T |author3=Krausz KW |author4=Sai Y |author5=Yang T |author6=Korzekwa KR |author7=Gonzalez FJ |author8=Gelboin HV | title =Use of inhibitory monoclonal antibodies to assess the contribution of cytochromes P450 to human drug metabolism| journal =]| volume =394| issue =2–3| pages =199–209| date =2000-04-14| doi =10.1016/S0014-2999(00)00079-0 | pmid =10771285}}</ref> There are five CYPs which are important in toluene metabolism, ], ], ], ], and ].<ref name=NakajimaBioPharm>{{cite journal| last =Nakajima| first =T|author2=Wang RS |author3=Elovaara E |author4=Gonzalez FJ |author5=Gelboin HV |author5-link=Harry Gelboin |author6=Raunio H |author7=Pelkonen O |author8=Vainio H |author9=Aoyama T | title =Toluene metabolism by cDNA-expressed human hepatic cytochrome P450| journal =]| volume =53| issue =3| pages =271–7| date =1997-02-07| doi =10.1016/S0006-2952(96)00652-1 | pmid =9065730}}</ref> The first four seem to be involved in the hydroxylation of toluene to benzyl alcohol. CYP2E1 seems to be the primary enzyme in the hydroxylation of toluene, accounting for roughly 44% of toluene metabolism;<ref name=Shou/> however, there is a great deal of ethnic variability, in the Finnish population for example the primary enzyme is ]. CYP2E1 catalyses the formation of benzyl alcohol and p-cresol,<ref name=Shou/><ref name=NakajimaBioPharm/> while CYP2B6 produces comparatively little ].<ref name=NakajimaBioPharm/> | ||
| Drug-discrimination studies using laboratory animals (Bowen et al., 1999) have shown that toluene can induce subjective effects similar to those of the psychedelic anesthetic ] (PCP), suggesting that toluene, like PCP, may block the NMDA receptor. It should be noted, however, that toluene failed to induce subjective effects similar to those of dizocilpine, another selective NMDA receptor blocker, in a similar drug-discrimination study (Shelton and Balster, 2004).<ref>https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3188822/</ref> | |||
| ⚫ | It is believed that in humans, benzyl alcohol is metabolised to ] by CYP rather than ];<ref name=Chapman>{{cite journal| last =Chapman| first =DE|author2=Moore TJ |author3=Michener SR |author4=Powis G | title =Metabolism and covalent binding of toluene by human and rat liver microsomal fractions and liver slices| journal =]| volume =18| issue =6| pages =929–36| date =November–December 1990| pmid =1981539 }}</ref> however, this belief does not appear to be universal.<ref name=ATSDR>{{cite book |author=Agency for Toxic Substances and Disease Registry |author-link=Agency for Toxic Substances and Disease Registry | title =Toxicological profile for toluene| publisher =Agency for Toxic Substances and Disease Registry|date=September 2000| location =], ]| url =http://www.atsdr.cdc.gov/toxprofiles/tp56.html| archive-url =https://web.archive.org/web/20010718214146/http://www.atsdr.cdc.gov/toxprofiles/tp56.html| url-status =dead| archive-date =July 18, 2001|oclc=47129207}}</ref><ref name=Kawamoto>{{cite journal| last =Kawamoto| first =T|author2=Matsuno K |author3=Kodama Y |author4=Murata K |author5=Matsuda S | title =ALDH2 polymorphism and biological monitoring of toluene| journal =]| volume =49| issue =5| pages =332–6| date =September–October 1994 |doi=10.1080/00039896.1994.9954983| pmid =7944563}}</ref> Benzaldehyde is in turn metabolised to ], primarily by mitochondrial ] (ALDH-2), while only a small percentage is metabolised by cytosolic ALDH-1.<ref name=Kawamoto/> | ||
| ⚫ | While a significant amount of ], 25% |
||
| ⚫ | Benzoic acid is metabolised to either benzoyl glucuronide or ].<ref name=ATSDR/><ref name=WHO>{{cite book| author=World Health Organization| author-link=World Health Organization| title =Environmental Health Criteria No. 52 (Toluene)| publisher =World Health Organization| year =1985| location =]| url =http://www.inchem.org/documents/ehc/ehc/ehc52.htm| isbn =978-92-4-154192-3 }}</ref> Benzoyl glucuronide is produced by the reaction of benzoic acid with glucuronic acid, which accounts for 10–20% of benzoic acid elimination. Hippuric acid is also known as benzoylglycine and is produced from benzoic acid in two steps: first benzoic acid is converted to benzoyl-CoA by the enzyme benzoyl-CoA synthase; then benzoyl-CoA is converted to hippuric acid by benzoyl-CoA:glycine N-acyltransferase.<ref name=Gregus>{{cite journal| last =Gregus| first =Z|author2=Fekete T |author3=Halászi E |author4=Klaassen CD | title =Lipoic acid impairs glycine conjugation of benzoic acid and renal excretion of benzoylglycine| journal =]| volume =24| issue =6| pages =682–8|date=June 1996| pmid =8781786}}</ref> Hippuric acid is the primary urinary metabolite of toluene.<ref name=ATSDR/> | ||
| ⚫ | It is believed that in humans, benzyl alcohol is metabolised to ] by CYP rather than ];<ref name=Chapman>{{cite journal| last =Chapman| first =DE| |
||
| ⚫ | Benzoic acid is metabolised to either benzoyl glucuronide or ].<ref name=ATSDR/><ref name=WHO>{{cite book| author= |
||
| ] | ] | ||
| Ring hydroxylation to cresols is a minor pathway in the metabolism of toluene. The majority of the cresol is excreted unchanged in urine; however, some of the p-cresol and ] is excreted as a conjugate. Studies in rats have shown that p-cresol is primarily conjugated with glucuronide to produce p-cresylglucuronide, though this may not be applicable to humans.<ref name=Lesaffer>{{cite journal| |
Ring hydroxylation to cresols is a minor pathway in the metabolism of toluene. The majority of the cresol is excreted unchanged in urine; however, some of the p-cresol and ] is excreted as a conjugate. Studies in rats have shown that p-cresol is primarily conjugated with glucuronide to produce p-cresylglucuronide, though this may not be applicable to humans.<ref name=Lesaffer>{{cite journal|vauthors=Lesaffer G, De Smet R, D'Heuvaert T, Belpaire FM, Lameire N, Vanholder R | title =Comparative kinetics of the uremic toxin p-cresol versus creatinine in rats with and without renal failure| journal =]| volume =64| issue =4| pages =1365–73|date=October 2003| doi =10.1046/j.1523-1755.2003.00228.x| pmid =12969155| doi-access =free}}</ref> o-cresol appears to be excreted mostly unchanged in urine or as the glucuronide or sulfate conjugate.<ref name=Wilkins-Haug>{{cite journal| last =Wilkins-Haug| first =L| title =Teratogen update: toluene| journal =]| volume =55| issue =2| pages =145–51|date=February 1997| doi =10.1002/(SICI)1096-9926(199702)55:2<145::AID-TERA5>3.0.CO;2-2| pmid =9143096}}</ref> There appears to be some dispute over whether ] is produced as a metabolite of toluene or not.<ref name=ATSDR/><ref name=Tassaneeyakul>{{cite journal| last =Tassaneeyakul| first =W|author2=Birkett DJ |author3=Edwards JW |author4=Veronese ME |author5=Tassaneeyakul W |author6=Tukey RH |author7=Miners JO | title =Human cytochrome P450 isoform specificity in the regioselective metabolism of toluene and o-, m- and p-xylene| journal =]| volume =276| issue =1| pages =101–8|date=January 1996| doi =10.1163/2211730x96x00063| pmid =8558417}}</ref> | ||
| ] | ] | ||
| Line 33: | Line 41: | ||
| When exposure to toluene occurs there is usually simultaneous exposure to several other chemicals.<ref name=ATSDR/> Often toluene exposure occurs in conjunction with ] and since they are to some degree metabolised by the same enzymes, the relative concentrations will determine their rate of elimination.<ref name=ATSDR/> Of course the longer it takes for toluene to be eliminated the more harm it is likely to do. | When exposure to toluene occurs there is usually simultaneous exposure to several other chemicals.<ref name=ATSDR/> Often toluene exposure occurs in conjunction with ] and since they are to some degree metabolised by the same enzymes, the relative concentrations will determine their rate of elimination.<ref name=ATSDR/> Of course the longer it takes for toluene to be eliminated the more harm it is likely to do. | ||
| The smoking and drinking habits of those exposed to toluene will partially determine the elimination of toluene. Studies have shown that even a modest amount of acute ethanol consumption can significantly decrease the distribution or elimination of toluene from the blood resulting in increased tissue exposure.<ref name=Wallen>{{cite journal| last =Wallen| first =M| |
The smoking and drinking habits of those exposed to toluene will partially determine the elimination of toluene. Studies have shown that even a modest amount of acute ethanol consumption can significantly decrease the distribution or elimination of toluene from the blood resulting in increased tissue exposure.<ref name=Wallen>{{cite journal| last =Wallen| first =M|author2=Näslund PH |author3=Nordqvist MB | title =The effects of ethanol on the kinetics of toluene in man| journal =]| volume =76| issue =3| pages =414–9|date=December 1984| doi =10.1016/0041-008X(84)90345-4| pmid =6506069}}</ref> Other studies have shown that chronic ethanol consumption can enhance toluene metabolism via the induction of CYP2E1.<ref name=NakajimaOccEnvHealth>{{cite journal| last =Nakajima| first =T|author2=Wang RS |author3=Murayama N | title =Immunochemical assessment of the influence of nutritional, physiological and environmental factors on the metabolism of toluene| journal =]| volume =65| issue =1 Supplement| pages =S127–30| year =1993| doi =10.1007/BF00381323| pmid =8406908| s2cid =41730461}}</ref> Smoking has been shown to enhance the elimination rate of toluene from the body, perhaps as a result of enzyme induction.<ref name=Hjelm>{{cite journal| last =Hjelm| first =EW|author2=Näslund PH |author3=Wallén M | title =Influence of cigarette smoking on the toxicokinetics of toluene in humans| journal =]| volume =25| issue =2| pages =155–63| year =1988| doi =10.1080/15287398809531197| pmid =3172270}}</ref> | ||
| The diet can also influence toluene elimination. Both a low-carbohydrate diet and fasting have been shown to induce CYP2E1 and as a result increase toluene metabolism.<ref name=NakajimaOccEnvHealth/> A low protein diet may decrease total CYP content and thereby reduce the elimination rate of the drug.<ref name=NakajimaOccEnvHealth/> | The diet can also influence toluene elimination. Both a low-carbohydrate diet and fasting have been shown to induce CYP2E1 and as a result increase toluene metabolism.<ref name=NakajimaOccEnvHealth/> A low protein diet may decrease total CYP content and thereby reduce the elimination rate of the drug.<ref name=NakajimaOccEnvHealth/> | ||
| ==Measure of exposure== | ==Measure of exposure== | ||
| ] has long been used as an indicator of toluene exposure;<ref name=Duydu>{{cite journal| last =Duydu| first =Y| |
] has long been used as an indicator of toluene exposure;<ref name=Duydu>{{cite journal| last =Duydu| first =Y|author2=Süzen S |author3=Erdem N |author4=Uysal H |author5=Vural N | title =Validation of hippuric acid as a biomarker of toluene exposure| journal =]| volume =63| issue =1| pages =1–8|date=July 1999| doi =10.1007/s001289900940| pmid =10423476| s2cid =41078122}}</ref> however, there appears to be some doubt about its validity.<ref name=AngererXII>{{cite journal| last =Angerer| first =J| title =Occupational chronic exposure to organic solvents. XII. O-cresol excretion after toluene exposure| journal =]| volume =56| issue =4| pages =323–8| year =1985| doi =10.1007/BF00405273| pmid =4066055| s2cid =11705704}}</ref> There is significant endogenous hippuric acid production by humans; which shows inter- and intra-individual variation influenced by factors such as diet, medical treatment, ] consumption, etc.<ref name=AngererXII/> This suggests that ] may be an unreliable indicator of toluene exposure.<ref name=AngererXII/><ref name=AngererXVI>{{cite journal| last =Angerer| first =J|author2=Krämer A | title =Occupational chronic exposure to organic solvents. XVI. Ambient and biological monitoring of workers exposed to toluene| journal =International Archives of Occupational and Environmental Health| volume =69| issue =2| pages =91–6| year =1997| doi =10.1007/s004200050121| pmid =9001914| s2cid =46369295}}</ref> It has been suggested that urinary hippuric acid, the traditional marker of toluene exposure is simply not sensitive enough to separate the exposed from the non-exposed.<ref name=InoueOccEnvHealthSeiji>{{cite journal| last =Inoue| first =O|author2=Seiji K |author3=Watanabe T |author4=Nakatsuka H |author5=Jin C |author6=Liu SJ |author7=Ikeda M | title =Effects of smoking and drinking on excretion of hippuric acid among toluene-exposed workers| journal =International Archives of Occupational and Environmental Health| volume =64| issue =6| pages =425–30| year =1993| doi =10.1007/BF00517948| pmid =8458658| s2cid =23244308}}</ref> This has led to the investigation of other metabolites as markers for toluene exposure.<ref name=AngererXVI/> | ||
| Urinary ] may be more reliable for the biomonitoring of toluene exposure because, unlike ], ] is not found at detectable levels in unexposed subjects.<ref name=AngererXVI/> o-Cresol may be a less sensitive marker of toluene exposure than hippuric acid.<ref name=InoueIndMed>{{cite journal| last =Inoue| first =O| |
Urinary ] may be more reliable for the biomonitoring of toluene exposure because, unlike ], ] is not found at detectable levels in unexposed subjects.<ref name=AngererXVI/> o-Cresol may be a less sensitive marker of toluene exposure than hippuric acid.<ref name=InoueIndMed>{{cite journal| last =Inoue| first =O|author2=Seiji K |author3=Watanabe T |author4=Chen Z |author5=Huang MY |author6=Xu XP |author7=Qiao X |author8=Ikeda M | title =Effects of smoking and drinking habits on urinary o-cresol excretion after occupational exposure to toluene vapor among Chinese workers| journal =]| volume =25| issue =5| pages =697–708|date=May 1994| doi =10.1002/ajim.4700250509| pmid =8030640}}</ref> o-Cresol excretion may be an unreliable method for measuring toluene exposure because o-cresol makes up <1% of total toluene elimination.<ref name=Duydu/> | ||
| ], a minor metabolite of toluene, is produced from ].<ref name=Laham>{{cite journal| last =Laham| first =S| |
], a minor metabolite of toluene, is produced from ].<ref name=Laham>{{cite journal| last =Laham| first =S|author2=Potvin M | title =Biological conversion of benzaldehyde to benzylmercapturic acid in the Sprague-Dawley rat| journal =]| volume =10| issue =3–4| pages =209–25| year =1987| doi =10.3109/01480548709042983| pmid =3428183}}</ref> In more recent years, studies have suggested the use of urinary ] as the best marker for toluene exposure, because: it is not detected in non-exposed subjects; it is more sensitive than ] at low concentrations; it is not affected by eating or drinking; it can detect toluene exposure down to approximately 15 ]; and it shows a better ] relationship with toluene than ] or ].<ref name=InoueToxLet>{{cite journal| last =Inoue| first =O|author2=Kanno E |author3=Kasai K |author4=Ukai H |author5=Okamoto S |author6=Ikeda M | title =Benzylmercapturic acid is superior to hippuric acid and o-cresol as a urinary marker of occupational exposure to toluene| journal =]| volume =147| issue =2| pages =177–86| date =2004-03-01| doi =10.1016/j.toxlet.2003.11.003 | pmid =14757321}}</ref><ref name=InoueOccEnvHealthKanno>{{cite journal| last =Inoue| first =O|author2=Kanno E |author3=Yusa T |author4=Kakizaki M |author5=Ukai H |author6=Okamoto S |author7=Higashikawa K |author8=Ikeda M | title =Urinary benzylmercapturic acid as a marker of occupational exposure to toluene| journal =International Archives of Occupational and Environmental Health| volume =75| issue =5| pages =341–7|date=June 2002| doi =10.1007/s00420-002-0322-8| pmid =11981673| s2cid =1992662}}</ref> | ||
| ==Effects of long |
==Effects of long-term exposure== | ||
| Serious adverse behavioural effects are often associated with toluene abuse related to the deliberate inhalation of solvents.<ref name=Miyagi>{{cite journal| last =Miyagi| first =Y| |
Serious adverse behavioural effects are often associated with chronic occupational exposure <ref>{{cite journal | author = Feldman RG, Ratner MH, Ptak T | date = May 1999 | title = Chronic toxic encephalopathy in a painter exposed to mixed solvents | journal = Environ Health Perspect | volume = 107 | issue = 5| pages = 417–22 | pmid = 10210698 | pmc = 1566426 | doi = 10.1289/ehp.99107417 }}</ref> and toluene abuse related to the deliberate ].<ref name=Miyagi>{{cite journal| last =Miyagi| first =Y|author2=Shima F |author3=Ishido K |author4=Yasutake T |author5=Kamikaseda K | title =Tremor induced by toluene misuse successfully treated by a Vim thalamotomy| journal =]| volume =66| issue =6| pages =794–6|date=June 1999| doi =10.1136/jnnp.66.6.794| pmid =10329759 | pmc=1736379}}</ref> Long-term toluene exposure is often associated with effects such as: ];<ref name=Urban>{{cite journal| last =Urban| first =P|author2=Lukáš E |author3=Pelclová D| title =Neurological and neurophysiological follow-up on workers with severe chronic exposure to toluene| journal =]| volume =P25| issue =s130| year =2003|display-authors=etal}}</ref> ] (VEP) abnormality;<ref name=Urban/> toxic ], ], ], and ];<ref name=Miyagi/><ref name=Urban/> ]; ]<ref>{{Cite journal|last1=Schäper|first1=Michael|last2=Seeber|first2=Andreas|last3=van Thriel|first3=Christoph|date=2008-01-01|title=The Effects of Toluene Plus Noise on Hearing Thresholds: An Evaluation Based on Repeated Measurements in the German Printing Industry|journal=International Journal of Occupational Medicine and Environmental Health|volume=21|issue=3|pages=191–200|doi=10.2478/v10001-008-0030-z|pmid=19042192|issn=1896-494X}}</ref><ref>{{Cite journal|last1=Yılmaz|first1=Omer Hinc|last2=Kos|first2=Mehmet|last3=Basturk|first3=Arzu|last4=Kesici|first4=Gulin Gokcen|last5=Unlu|first5=Ilhan|date=2014-11-01|title=A comparison of the effects of solvent and noise exposure on hearing, together and separately|journal=Noise and Health|language=en|volume=16|issue=73|pages=410–5|doi=10.4103/1463-1741.144422|issn=1463-1741|pmid=25387537|doi-access=free|hdl=20.500.12684/2335|hdl-access=free}}</ref> and ].<ref name=Miyagi/> | ||
| The neurotoxic effects of long |
The neurotoxic effects of long-term use (in particular ]) of toluene may cause postural tremors by downregulating ]s within the ].<ref name=Miyagi/> Treatment with ] agonists such as ] provide some relief from toluene-induced tremor and ].<ref name=Miyagi/> An alternative to drug treatment is ].<ref name=Miyagi/> The tremors associated with toluene misuse do not seem to be a transient symptom, but an irreversible and progressive symptom which continues after solvent abuse has been discontinued.<ref name=Miyagi/> | ||
| There is some evidence that low |
There is some evidence that low-level toluene exposure may cause disruption in the differentiation of ] precursor cells.<ref name=Yamaguchi>{{cite journal| last =Yamaguchi| first =H|author2=Kidachi Y |author3=Ryoyama K | title =Toluene at environmentally relevant low levels disrupts differentiation of astrocyte precursor cells| journal =Archives of Environmental Health| volume =57| issue =3| pages =232–8| date =May–June 2002| doi =10.1080/00039890209602942| pmid =12507177| s2cid =22967797}}</ref> This does not appear to be a major hazard to adults; however, exposure of pregnant women to toluene during critical stages of fetal development could cause serious disruption to neuronal development.<ref name=Yamaguchi/> | ||
| ==References== | ==References== | ||
| {{ |
{{Reflist}} | ||
| ==External links== | == External links == | ||
| {{Medical resources | |||
| ⚫ | * U.S. Department of Health and Human Services (public domain) | ||
| | DiseasesDB = 31145 | |||
| | ICD10 = T52.2 | |||
| | ICD9 = {{ICD9|982.0}} | |||
| | ICDO = | |||
| | OMIM = | |||
| | MedlinePlus = | |||
| | eMedicineSubj = article | |||
| ⚫ | | eMedicineTopic = 818939 | ||
| | MeshID = | |||
| }} | |||
| ⚫ | * U.S. Department of Health and Human Services (public domain) | ||
| {{Psychoactive substance use}} | {{Psychoactive substance use}} | ||
Latest revision as of 06:11, 30 December 2024
Medical condition| Toluene toxicity | |
|---|---|
 | |
| Chemical structure of toluene | |
| Specialty | Emergency medicine |
Toluene toxicity refers to the harmful effects caused by toluene on the body.
Metabolism in humans
While a significant amount of toluene, 25%–40%, is exhaled unchanged via the lungs, a greater proportion is metabolised and excreted via other pathways. The primary route of toluene metabolism is by hydroxylation to benzyl alcohol by members of the cytochrome P450 (CYP) superfamily. There are five CYPs which are important in toluene metabolism, CYP1A2, CYP2B6, CYP2E1, CYP2C8, and CYP1A1. The first four seem to be involved in the hydroxylation of toluene to benzyl alcohol. CYP2E1 seems to be the primary enzyme in the hydroxylation of toluene, accounting for roughly 44% of toluene metabolism; however, there is a great deal of ethnic variability, in the Finnish population for example the primary enzyme is CYP2B6. CYP2E1 catalyses the formation of benzyl alcohol and p-cresol, while CYP2B6 produces comparatively little p-cresol.
It is believed that in humans, benzyl alcohol is metabolised to benzaldehyde by CYP rather than alcohol dehydrogenase; however, this belief does not appear to be universal. Benzaldehyde is in turn metabolised to benzoic acid, primarily by mitochondrial aldehyde dehydrogenase-2 (ALDH-2), while only a small percentage is metabolised by cytosolic ALDH-1.
Benzoic acid is metabolised to either benzoyl glucuronide or hippuric acid. Benzoyl glucuronide is produced by the reaction of benzoic acid with glucuronic acid, which accounts for 10–20% of benzoic acid elimination. Hippuric acid is also known as benzoylglycine and is produced from benzoic acid in two steps: first benzoic acid is converted to benzoyl-CoA by the enzyme benzoyl-CoA synthase; then benzoyl-CoA is converted to hippuric acid by benzoyl-CoA:glycine N-acyltransferase. Hippuric acid is the primary urinary metabolite of toluene.

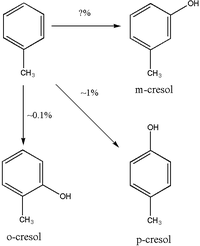
Ring hydroxylation to cresols is a minor pathway in the metabolism of toluene. The majority of the cresol is excreted unchanged in urine; however, some of the p-cresol and o-cresol is excreted as a conjugate. Studies in rats have shown that p-cresol is primarily conjugated with glucuronide to produce p-cresylglucuronide, though this may not be applicable to humans. o-cresol appears to be excreted mostly unchanged in urine or as the glucuronide or sulfate conjugate. There appears to be some dispute over whether m-cresol is produced as a metabolite of toluene or not.
Environmental influences
When exposure to toluene occurs there is usually simultaneous exposure to several other chemicals. Often toluene exposure occurs in conjunction with benzene and since they are to some degree metabolised by the same enzymes, the relative concentrations will determine their rate of elimination. Of course the longer it takes for toluene to be eliminated the more harm it is likely to do.
The smoking and drinking habits of those exposed to toluene will partially determine the elimination of toluene. Studies have shown that even a modest amount of acute ethanol consumption can significantly decrease the distribution or elimination of toluene from the blood resulting in increased tissue exposure. Other studies have shown that chronic ethanol consumption can enhance toluene metabolism via the induction of CYP2E1. Smoking has been shown to enhance the elimination rate of toluene from the body, perhaps as a result of enzyme induction.
The diet can also influence toluene elimination. Both a low-carbohydrate diet and fasting have been shown to induce CYP2E1 and as a result increase toluene metabolism. A low protein diet may decrease total CYP content and thereby reduce the elimination rate of the drug.
Measure of exposure
Hippuric acid has long been used as an indicator of toluene exposure; however, there appears to be some doubt about its validity. There is significant endogenous hippuric acid production by humans; which shows inter- and intra-individual variation influenced by factors such as diet, medical treatment, alcohol consumption, etc. This suggests that hippuric acid may be an unreliable indicator of toluene exposure. It has been suggested that urinary hippuric acid, the traditional marker of toluene exposure is simply not sensitive enough to separate the exposed from the non-exposed. This has led to the investigation of other metabolites as markers for toluene exposure.
Urinary o-cresol may be more reliable for the biomonitoring of toluene exposure because, unlike hippuric acid, o-cresol is not found at detectable levels in unexposed subjects. o-Cresol may be a less sensitive marker of toluene exposure than hippuric acid. o-Cresol excretion may be an unreliable method for measuring toluene exposure because o-cresol makes up <1% of total toluene elimination.
Benzylmercapturic acid, a minor metabolite of toluene, is produced from benzaldehyde. In more recent years, studies have suggested the use of urinary benzylmercapturic acid as the best marker for toluene exposure, because: it is not detected in non-exposed subjects; it is more sensitive than hippuric acid at low concentrations; it is not affected by eating or drinking; it can detect toluene exposure down to approximately 15 ppm; and it shows a better quantitative relationship with toluene than hippuric acid or o-cresol.
Effects of long-term exposure
Serious adverse behavioural effects are often associated with chronic occupational exposure and toluene abuse related to the deliberate inhalation of solvents. Long-term toluene exposure is often associated with effects such as: psychoorganic syndrome; visual evoked potential (VEP) abnormality; toxic polyneuropathy, cerebellar, cognitive, and pyramidal dysfunctions; optic atrophy; hearing disorders and brain lesions.
The neurotoxic effects of long-term use (in particular repeated withdrawals) of toluene may cause postural tremors by downregulating GABA receptors within the cerebellar cortex. Treatment with GABA agonists such as benzodiazepines provide some relief from toluene-induced tremor and ataxia. An alternative to drug treatment is ventral intermediate nucleus (vim) thalamotomy. The tremors associated with toluene misuse do not seem to be a transient symptom, but an irreversible and progressive symptom which continues after solvent abuse has been discontinued.
There is some evidence that low-level toluene exposure may cause disruption in the differentiation of astrocyte precursor cells. This does not appear to be a major hazard to adults; however, exposure of pregnant women to toluene during critical stages of fetal development could cause serious disruption to neuronal development.
References
- ^ Shou, M; Lu T; Krausz KW; Sai Y; Yang T; Korzekwa KR; Gonzalez FJ; Gelboin HV (2000-04-14). "Use of inhibitory monoclonal antibodies to assess the contribution of cytochromes P450 to human drug metabolism". European Journal of Pharmacology. 394 (2–3): 199–209. doi:10.1016/S0014-2999(00)00079-0. PMID 10771285.
- ^ Nakajima, T; Wang RS; Elovaara E; Gonzalez FJ; Gelboin HV; Raunio H; Pelkonen O; Vainio H; Aoyama T (1997-02-07). "Toluene metabolism by cDNA-expressed human hepatic cytochrome P450". Biochemical Pharmacology. 53 (3): 271–7. doi:10.1016/S0006-2952(96)00652-1. PMID 9065730.
- Chapman, DE; Moore TJ; Michener SR; Powis G (November–December 1990). "Metabolism and covalent binding of toluene by human and rat liver microsomal fractions and liver slices". Drug Metabolism and Disposition. 18 (6): 929–36. PMID 1981539.
- ^ Agency for Toxic Substances and Disease Registry (September 2000). Toxicological profile for toluene. Atlanta, GA: Agency for Toxic Substances and Disease Registry. OCLC 47129207. Archived from the original on July 18, 2001.
- ^ Kawamoto, T; Matsuno K; Kodama Y; Murata K; Matsuda S (September–October 1994). "ALDH2 polymorphism and biological monitoring of toluene". Archives of Environmental Health. 49 (5): 332–6. doi:10.1080/00039896.1994.9954983. PMID 7944563.
- World Health Organization (1985). Environmental Health Criteria No. 52 (Toluene). Geneva: World Health Organization. ISBN 978-92-4-154192-3.
- Gregus, Z; Fekete T; Halászi E; Klaassen CD (June 1996). "Lipoic acid impairs glycine conjugation of benzoic acid and renal excretion of benzoylglycine". Drug Metabolism and Disposition. 24 (6): 682–8. PMID 8781786.
- Lesaffer G, De Smet R, D'Heuvaert T, Belpaire FM, Lameire N, Vanholder R (October 2003). "Comparative kinetics of the uremic toxin p-cresol versus creatinine in rats with and without renal failure". Kidney International. 64 (4): 1365–73. doi:10.1046/j.1523-1755.2003.00228.x. PMID 12969155.
- Wilkins-Haug, L (February 1997). "Teratogen update: toluene". Teratology. 55 (2): 145–51. doi:10.1002/(SICI)1096-9926(199702)55:2<145::AID-TERA5>3.0.CO;2-2. PMID 9143096.
- Tassaneeyakul, W; Birkett DJ; Edwards JW; Veronese ME; Tassaneeyakul W; Tukey RH; Miners JO (January 1996). "Human cytochrome P450 isoform specificity in the regioselective metabolism of toluene and o-, m- and p-xylene". Journal of Pharmacology and Experimental Therapeutics. 276 (1): 101–8. doi:10.1163/2211730x96x00063. PMID 8558417.
- Wallen, M; Näslund PH; Nordqvist MB (December 1984). "The effects of ethanol on the kinetics of toluene in man". Toxicology and Applied Pharmacology. 76 (3): 414–9. doi:10.1016/0041-008X(84)90345-4. PMID 6506069.
- ^ Nakajima, T; Wang RS; Murayama N (1993). "Immunochemical assessment of the influence of nutritional, physiological and environmental factors on the metabolism of toluene". International Archives of Occupational and Environmental Health. 65 (1 Supplement): S127–30. doi:10.1007/BF00381323. PMID 8406908. S2CID 41730461.
- Hjelm, EW; Näslund PH; Wallén M (1988). "Influence of cigarette smoking on the toxicokinetics of toluene in humans". Journal of Toxicology and Environmental Health. 25 (2): 155–63. doi:10.1080/15287398809531197. PMID 3172270.
- ^ Duydu, Y; Süzen S; Erdem N; Uysal H; Vural N (July 1999). "Validation of hippuric acid as a biomarker of toluene exposure". Bulletin of Environmental Contamination and Toxicology. 63 (1): 1–8. doi:10.1007/s001289900940. PMID 10423476. S2CID 41078122.
- ^ Angerer, J (1985). "Occupational chronic exposure to organic solvents. XII. O-cresol excretion after toluene exposure". International Archives of Occupational and Environmental Health. 56 (4): 323–8. doi:10.1007/BF00405273. PMID 4066055. S2CID 11705704.
- ^ Angerer, J; Krämer A (1997). "Occupational chronic exposure to organic solvents. XVI. Ambient and biological monitoring of workers exposed to toluene". International Archives of Occupational and Environmental Health. 69 (2): 91–6. doi:10.1007/s004200050121. PMID 9001914. S2CID 46369295.
- Inoue, O; Seiji K; Watanabe T; Nakatsuka H; Jin C; Liu SJ; Ikeda M (1993). "Effects of smoking and drinking on excretion of hippuric acid among toluene-exposed workers". International Archives of Occupational and Environmental Health. 64 (6): 425–30. doi:10.1007/BF00517948. PMID 8458658. S2CID 23244308.
- Inoue, O; Seiji K; Watanabe T; Chen Z; Huang MY; Xu XP; Qiao X; Ikeda M (May 1994). "Effects of smoking and drinking habits on urinary o-cresol excretion after occupational exposure to toluene vapor among Chinese workers". American Journal of Industrial Medicine. 25 (5): 697–708. doi:10.1002/ajim.4700250509. PMID 8030640.
- Laham, S; Potvin M (1987). "Biological conversion of benzaldehyde to benzylmercapturic acid in the Sprague-Dawley rat". Drug and Chemical Toxicology. 10 (3–4): 209–25. doi:10.3109/01480548709042983. PMID 3428183.
- Inoue, O; Kanno E; Kasai K; Ukai H; Okamoto S; Ikeda M (2004-03-01). "Benzylmercapturic acid is superior to hippuric acid and o-cresol as a urinary marker of occupational exposure to toluene". Toxicology Letters. 147 (2): 177–86. doi:10.1016/j.toxlet.2003.11.003. PMID 14757321.
- Inoue, O; Kanno E; Yusa T; Kakizaki M; Ukai H; Okamoto S; Higashikawa K; Ikeda M (June 2002). "Urinary benzylmercapturic acid as a marker of occupational exposure to toluene". International Archives of Occupational and Environmental Health. 75 (5): 341–7. doi:10.1007/s00420-002-0322-8. PMID 11981673. S2CID 1992662.
- Feldman RG, Ratner MH, Ptak T (May 1999). "Chronic toxic encephalopathy in a painter exposed to mixed solvents". Environ Health Perspect. 107 (5): 417–22. doi:10.1289/ehp.99107417. PMC 1566426. PMID 10210698.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Miyagi, Y; Shima F; Ishido K; Yasutake T; Kamikaseda K (June 1999). "Tremor induced by toluene misuse successfully treated by a Vim thalamotomy". Journal of Neurology, Neurosurgery, and Psychiatry. 66 (6): 794–6. doi:10.1136/jnnp.66.6.794. PMC 1736379. PMID 10329759.
- ^ Urban, P; Lukáš E; Pelclová D; et al. (2003). "Neurological and neurophysiological follow-up on workers with severe chronic exposure to toluene". Neurotoxicity. P25 (s130).
- Schäper, Michael; Seeber, Andreas; van Thriel, Christoph (2008-01-01). "The Effects of Toluene Plus Noise on Hearing Thresholds: An Evaluation Based on Repeated Measurements in the German Printing Industry". International Journal of Occupational Medicine and Environmental Health. 21 (3): 191–200. doi:10.2478/v10001-008-0030-z. ISSN 1896-494X. PMID 19042192.
- Yılmaz, Omer Hinc; Kos, Mehmet; Basturk, Arzu; Kesici, Gulin Gokcen; Unlu, Ilhan (2014-11-01). "A comparison of the effects of solvent and noise exposure on hearing, together and separately". Noise and Health. 16 (73): 410–5. doi:10.4103/1463-1741.144422. hdl:20.500.12684/2335. ISSN 1463-1741. PMID 25387537.
- ^ Yamaguchi, H; Kidachi Y; Ryoyama K (May–June 2002). "Toluene at environmentally relevant low levels disrupts differentiation of astrocyte precursor cells". Archives of Environmental Health. 57 (3): 232–8. doi:10.1080/00039890209602942. PMID 12507177. S2CID 22967797.
External links
| Classification | D |
|---|---|
| External resources |
- ATSDR - Case Studies in Environmental Medicine: Toluene Toxicity U.S. Department of Health and Human Services (public domain)
| Psychoactive substance-related disorders | |||||
|---|---|---|---|---|---|
| General | |||||
| Combined substance use |
| ||||
| Alcohol |
| ||||
| Caffeine | |||||
| Cannabis | |||||
| Cocaine | |||||
| Hallucinogen | |||||
| Nicotine | |||||
| Opioids |
| ||||
| Sedative / hypnotic | |||||
| Stimulants | |||||
| Volatile solvent | |||||
| Related | |||||
| Inorganic |
| ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Organic |
| ||||||||||||
| Pharmaceutical |
| ||||||||||||
| Biological |
| ||||||||||||
| Miscellaneous | |||||||||||||
| |||||||||||||