| Revision as of 00:27, 18 June 2011 editVCalvoP (talk | contribs)59 editsNo edit summary← Previous edit | Revision as of 01:09, 18 June 2011 edit undoVCalvoP (talk | contribs)59 editsNo edit summaryNext edit → | ||
| Line 57: | Line 57: | ||
| The most cited path to alkyl iodides include the typical halogenation using HI solution and 2-propanol, but the regioselectivity is usually improved using PI3 prepared in-situ. | The most cited path to alkyl iodides include the typical halogenation using HI solution and 2-propanol, but the regioselectivity is usually improved using PI3 prepared in-situ. | ||
| An alternative preparation involves halide exchange using potassium iodide (KI: 0.25 mol) dissolved in dry dimethylcetone. A solution of 0.2 mol of 2-propyl bromide in dry acetone (250 mL) is allowed to react at room temperature with the KI freshly prepared in acetone. A fine precipitate of potassium bromide forms immediately. The reaction is allowed to proceed under reflux and fractional distillation is required to boil of the dimethylcetone and unreacted 2-propanol. The 2-propyl iodide boils of between 88 to 89 °C. Good to moderate yield ( |
An alternative preparation involves halide exchange using potassium iodide (KI: 0.25 mol) dissolved in dry dimethylcetone. A solution of 0.2 mol of 2-propyl bromide in dry acetone (250 mL) is allowed to react at room temperature with the KI freshly prepared in acetone. A fine precipitate of potassium bromide forms immediately. The reaction is allowed to proceed under reflux and fractional distillation is required to boil of the dimethylcetone and unreacted 2-propanol. The 2-propyl iodide boils of between 88 to 89 °C. Good to moderate yield (70%) is obtained within 60 minutes. | ||
| The reaction is believed to occur via S<sub>N</sub>2 mechanism, therefore an inversion of configuration is expected for chiral halides. <ref>Textbook of Practical Organic Chemistry, 5th Edition, Prentice Hall, 1989</ref> | The reaction is believed to occur via S<sub>N</sub>2 mechanism, therefore an inversion of configuration is expected for chiral halides. This reaction path is preferable for the synthesis of 1-propyl iodide. | ||
| <ref>Textbook of Practical Organic Chemistry, 5th Edition, Prentice Hall, 1989</ref> | |||
| ==References== | ==References== | ||
Revision as of 01:09, 18 June 2011

| |
| |
| Names | |
|---|---|
| IUPAC name 2-iodopropane | |
| Other names iododimethylmethane, isopropyl iodide, 2-propyl iodide, sec-propyl iodide | |
| Identifiers | |
| CAS Number | |
| 3D model (JSmol) | |
| ECHA InfoCard | 100.000.782 |
| RTECS number |
|
| CompTox Dashboard (EPA) | |
SMILES
| |
| Properties | |
| Chemical formula | C3H7I |
| Molar mass | 169.99 |
| Appearance | Colourless liquid |
| Density | 1.703 |
| Melting point | −90.0 °C (−130.0 °F; 183.2 K) |
| Boiling point | 89.5 °C (193.1 °F; 362.6 K) |
| Solubility in water | 0.14 g/100 ml at 12.5 °C |
| Solubility in ethanol | fully miscible |
| Solubility in diethyl ether | fully miscible |
| Solubility in chloroform | fully miscible |
| Solubility in benzene | fully miscible |
| Refractive index (nD) | 1.4997 |
| Viscosity | 8.841 cP at 0 °C 6.971 cP at 20 °C |
| Hazards | |
| Occupational safety and health (OHS/OSH): | |
| Main hazards | Possible carcinogen. Harmful if swallowed, inhaled and in contact with skin. Eye, respiratory and mucous membrane irritant. |
| Flash point | 42 °C |
| Related compounds | |
| Except where otherwise noted, data are given for materials in their standard state (at 25 °C , 100 kPa).
| |
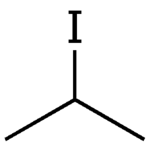
Isopropyl iodide (also iododimethylmethane, 2-propyl iodide, sec-propyl iodide or 2-iodopropane) is a colorless, flammable chemical compound. It has the chemical formula C3H7I and is prepared by distilling isopropyl alcohol with hydrogen iodide, or with glycerol, iodine, and phosphorus.
The most cited path to alkyl iodides include the typical halogenation using HI solution and 2-propanol, but the regioselectivity is usually improved using PI3 prepared in-situ. An alternative preparation involves halide exchange using potassium iodide (KI: 0.25 mol) dissolved in dry dimethylcetone. A solution of 0.2 mol of 2-propyl bromide in dry acetone (250 mL) is allowed to react at room temperature with the KI freshly prepared in acetone. A fine precipitate of potassium bromide forms immediately. The reaction is allowed to proceed under reflux and fractional distillation is required to boil of the dimethylcetone and unreacted 2-propanol. The 2-propyl iodide boils of between 88 to 89 °C. Good to moderate yield (70%) is obtained within 60 minutes. The reaction is believed to occur via SN2 mechanism, therefore an inversion of configuration is expected for chiral halides. This reaction path is preferable for the synthesis of 1-propyl iodide.
References
- Merck Index of Chemicals and Drugs, 9th ed., monograph 5074
- Textbook of Practical Organic Chemistry, 5th Edition, Prentice Hall, 1989
This article about an organic halide is a stub. You can help Misplaced Pages by expanding it. |