| |

| |
| Names | |
|---|---|
| Other names Kelocyanor | |
| Identifiers | |
| CAS Number | |
| 3D model (JSmol) | |
| ChemSpider | |
| ECHA InfoCard | 100.048.227 |
| PubChem CID | |
| UNII | |
| CompTox Dashboard (EPA) | |
InChI
| |
SMILES
| |
| Properties | |
| Chemical formula | C10H12Co2N2O8.6H2O |
| Molar mass | 406.08 g/mol 514.18 g/mol (hexahydrate) |
| Except where otherwise noted, data are given for materials in their standard state (at 25 °C , 100 kPa).
| |
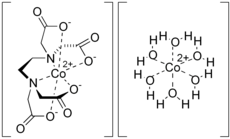
Dicobalt edetate is the coordination compound with the approximate formula Co2(EDTA)(H2O)6. It is a derivative of the (non-natural) amino acid ethylenediaminetetraacetate.
Solutions of this solid have been used in Europe as an antidote to cyanide poisoning, with the trade name Kelocyanor. However, dicobalt edetate is itself toxic, particularly if there is not enough cyanide in the blood to react with it, so it is only suitable for serious cases of poisoning. In the past, workplaces in the UK that handle cyanide were recommended to keep a 'Kelocyanor kit', to be taken to the hospital along with the patient, but this practice has been discontinued since it may mislead doctors into using the antidote unnecessarily.
Structure
The compound is polymeric in the crystalline form. Half of the Co ions are bound to the EDTA and the other Co ions are bound to four water ligands as well as carboxylate ligands on the entity. In aqueous solution, depolymerization occurs to give and ions, each of which is kinetically labile and has a high affinity for cyanide.
Related compounds
Oxidation of gives , which is so kinetically inert that it can be resolved optically.
References
- Pickering WG (December 1985). "Cyanide toxicity and the hazards of dicobalt edetate". Br Med J (Clin Res Ed). 291 (6509): 1644. doi:10.1136/bmj.291.6509.1644-a. PMC 1418389. PMID 2866807.
- "Cyanide poisoning – New recommendations on first aid treatment". Health and Safety Executive. Archived from the original on 20 October 2009.
- E. F. K. Mccandlish, T. K. Michael; Rose, N. J.; Neal, J. A.; Lingafelter, E. C.; Rose, N. J. (1978). "Comparison of the Structures and Aqueous Solutions of Cobalt(II) and Cobalt(II)". Inorg. Chem. 17 (6): 1383–94. doi:10.1021/ic50184a001.
- Dwyer, F. P.; Garvan, F. L. (1960). "Resolution of the Ethylenediaminetetraacetatocobaltate(III) Ion". Inorganic Syntheses. Vol. VI. pp. 192–4. doi:10.1002/9780470132371.ch61. ISBN 978-0-470-13237-1.
{{cite book}}:|journal=ignored (help)