| This article may need to be rewritten to comply with Misplaced Pages's quality standards. You can help. The talk page may contain suggestions. (March 2020) |
| |
| Names | |
|---|---|
| Preferred IUPAC name (3R,4R)-3,4-Bisoxolan-2-one | |
| Other names (3R,4R)-3,4-Bis(1,3-benzodioxol-5-ylmethyl)dihydro-2(3H)-furanone | |
| Identifiers | |
| CAS Number | |
| 3D model (JSmol) | |
| ChEMBL | |
| ChemSpider | |
| PubChem CID | |
| CompTox Dashboard (EPA) | |
InChI
| |
SMILES
| |
| Properties | |
| Chemical formula | C20H18O6 |
| Molar mass | 354.358 g·mol |
| Except where otherwise noted, data are given for materials in their standard state (at 25 °C , 100 kPa).
| |
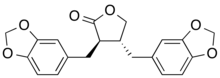
Hinokinin is a dibenzylbutyrolactone lignan, derived from various species of plants. It is a potential antichagonistic agent. In vitro, it has been shown to have potential neuroprotective effects as well as anti-inflammatory, anti-tumor, antiviral and antifungal properties.
Hinokinin was isolated for the first time by Yoshiki and Ishiguro in 1933 from hinoki wood.
Chemical properties
Hinokinin is a colourless crystalline compound.
It can be isolated from various species of Chamaecyparis, Zanthoxylum, Phyllanthus, Aristolochia, Piper, Virola, Linum and Bursera. It is also synthesised from pinoresinol.
Biological effects
Cytotoxic actions
Hinokinin has shown to induce apoptosis and promote antitumor actions on various cancer cell lines in vitro.
Anti-inflammatory actions
Hinokinin has been shown to inhibit the generation of superoxide molecules by neutrophils and also decreases elastase secretion from neutrophils. It has also shown to reduce LPS induced nitric oxide production from macrophages.The anti-inflammatory property of hinokinin is mediated by the NF-kB signalling mechanism.
Anti-parasitic actions
Hinokinin has been shown to be an antitrypanosomal agent. Its use as a treatment for trypanosomiasis is still being researched.
Anti-viral actions
It has shown significant antiviral activity against human hepatitis B virus, HIV and SARS-CoV.
See also
References
- ^ Marcotullio, Maria; Pelosi, Azzurra; Curini, Massimo (2014-09-17). "Hinokinin, an Emerging Bioactive Lignan". Molecules. 19 (9): 14862–14878. doi:10.3390/molecules190914862. ISSN 1420-3049. PMC 6271885. PMID 25232707.
- Timple, Julie Marie V.; Magalhães, Lizandra Guidi; Souza Rezende, Karen Cristina; Pereira, Ana Carolina; Cunha, Wilson Roberto; Andrade e Silva, Márcio Luis; Mortensen, Ole Valente; Fontana, Andréia C. K. (2013-10-10). "The Lignan (−)-Hinokinin Displays Modulatory Effects on Human Monoamine and GABA Transporter Activities". Journal of Natural Products. 76 (10): 1889–1895. doi:10.1021/np400452n. ISSN 0163-3864. PMID 24112084.
- Zhou, Qi-Long; Wang, Hui-Jing; Tang, Pei; Song, Hao; Qin, Yong (October 2015). "Total Synthesis of Lignan Lactone (–)-Hinokinin". Natural Products and Bioprospecting. 5 (5): 255–261. doi:10.1007/s13659-015-0073-3. ISSN 2192-2195. PMC 4607678. PMID 26458924.
- Yoshiki, Y.; Ishiguro, T. (1933). "Ueber die kristallisierten Bestandteile des Hinokiöls". Yakugaku Zasshi. 53 (2): 73–151. doi:10.1248/yakushi1881.53.2_73. ISSN 0031-6903.
- Cao, Xue-li; Xu, Jing; Bai, Ge; Zhang, Hong; Liu, Yan; Xiang, Jun-feng; Tang, Ya-lin (June 2013). "Isolation of anti-tumor compounds from the stem bark of Zanthoxylum ailanthoides Sieb. & Zucc. by silica gel column and counter-current chromatography". Journal of Chromatography B. 929: 6–10. doi:10.1016/j.jchromb.2013.04.006. ISSN 1570-0232. PMID 23660246.
- Chen, JJ; Chung, CY; Hwang, TL; Chen, JF (July 2009). "Amides and Benzenoids from Zanthoxylum ailanthoides with Inhibitory Activity on Superoxide Generation and Elastase Release by Neutrophils". Planta Medica. 75 (9). doi:10.1055/s-0029-1234991. ISSN 0032-0943.
- Desai, Dattatraya C.; Jacob, Jeenu; Almeida, Asha; Kshirsagar, Rajendra; Manju, S.L. (2014-05-23). "Isolation, structural elucidation and anti-inflammatory activity of astragalin, ( − )hinokinin, aristolactam I and aristolochic acids (I & II) fromAristolochia indica". Natural Product Research. 28 (17): 1413–1417. doi:10.1080/14786419.2014.905563. ISSN 1478-6419. PMID 24854204. S2CID 20632496.
- Saraiva, Juliana; Lira, Ana Amélia Moreira; Esperandim, Viviane Rodrigues; da Silva Ferreira, Daniele; Ferraudo, Antônio Sérgio; Bastos, Jairo Kenupp; e Silva, Márcio Luís Andrade; de Gaitani, Cristiane Masetto; de Albuquerque, Sérgio; Marchetti, Juliana Maldonado (2010-01-28). "(−)−Hinokinin-loaded poly(D,L-lactide-co-glycolide) microparticles for Chagas disease". Parasitology Research. 106 (3): 703–708. doi:10.1007/s00436-010-1725-1. ISSN 0932-0113. PMID 20107838. S2CID 31990739.
- Wen, Chih-Chun; Kuo, Yueh-Hsiung; Jan, Jia-Tsrong; Liang, Po-Huang; Wang, Sheng-Yang; Liu, Hong-Gi; Lee, Ching-Kuo; Chang, Shang-Tzen; Kuo, Chih-Jung; Lee, Shoei-Sheng; Hou, Chia-Chung (August 2007). "Specific Plant Terpenoids and Lignoids Possess Potent Antiviral Activities against Severe Acute Respiratory Syndrome Coronavirus" (PDF). Journal of Medicinal Chemistry. 50 (17): 4087–4095. doi:10.1021/jm070295s. ISSN 0022-2623. PMID 17663539.