This is an old revision of this page, as edited by CheMoBot (talk | contribs) at 00:06, 7 September 2011 (Updating {{chembox}} (no changed fields - added verified revid - updated 'ChemSpiderID_Ref', 'DrugBank_Ref', 'UNII_Ref', 'ChEMBL_Ref', 'ChEBI_Ref', 'KEGG_Ref', 'StdInChI_Ref', 'StdInChIKey_Ref', 'ChEBI_Ref') per [[WP:CHEMVALID|Chem/Drugbox validation). The present address (URL) is a permanent link to this revision, which may differ significantly from the current revision.
Revision as of 00:06, 7 September 2011 by CheMoBot (talk | contribs) (Updating {{chembox}} (no changed fields - added verified revid - updated 'ChemSpiderID_Ref', 'DrugBank_Ref', 'UNII_Ref', 'ChEMBL_Ref', 'ChEBI_Ref', 'KEGG_Ref', 'StdInChI_Ref', 'StdInChIKey_Ref', 'ChEBI_Ref') per [[WP:CHEMVALID|Chem/Drugbox validation)(diff) ← Previous revision | Latest revision (diff) | Newer revision → (diff)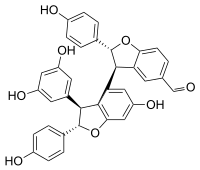
| |
| Names | |
|---|---|
| IUPAC name (2R,2′S,3R,3′S)-3′-(3,5-Dihydroxyphenyl)-6′-hydroxy-2,2′-bis(4-hydroxyphenyl)-2,2′,3,3′-tetrahydro--5-carbaldehyde | |
| Other names (-)-Viniferal | |
| Identifiers | |
| CAS Number | |
| 3D model (JSmol) | |
| ChemSpider | |
| CompTox Dashboard (EPA) | |
InChI
| |
SMILES
| |
| Properties | |
| Chemical formula | C35H26O8 |
| Molar mass | 574.585 g·mol |
| Except where otherwise noted, data are given for materials in their standard state (at 25 °C , 100 kPa).
| |
Viniferal is a hydroxystilbenoid with an aldehyde group found in Vitis vinifera (grapevine).
References
- Ito, J (1996). "Absolute structures of new hydroxystilbenoids, vitisin C and viniferal, from Vitis vinifera 'Kyohou'". Tetrahedron. 52 (30): 9991. doi:10.1016/0040-4020(96)00543-1.
External links
| Type of stilbenoids (molecules with a C6-C2-C6 backbone) | |
|---|---|
| Types |
|