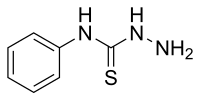
| |
| Names | |
|---|---|
| Preferred IUPAC name N-Phenylhydrazinecarbothioamide | |
| Other names N-Phenyl-hydrazinecarbothioamide, USAF-EK-5426, USAF-EL-45 | |
| Identifiers | |
| CAS Number | |
| 3D model (JSmol) | |
| ChemSpider | |
| ECHA InfoCard | 100.023.935 |
| PubChem CID | |
| UNII | |
| CompTox Dashboard (EPA) | |
InChI
| |
SMILES
| |
| Properties | |
| Chemical formula | C7H9N3S |
| Molar mass | 167.23 g·mol |
| Appearance | White to light yellow crystalline powder |
| Melting point | 138 to 141 °C (280 to 286 °F; 411 to 414 K) |
| Boiling point | 308 °C (586 °F; 581 K) |
| Except where otherwise noted, data are given for materials in their standard state (at 25 °C , 100 kPa).
| |
4-Phenylthiosemicarbazide (4-PTSC) is a thiosemicarbazide used as an agricultural chemical and pesticide. It also possesses antibacterial properties attributed to electron delocalization in the thiosemicarbazide moiety.
References
- CID 730679 from PubChem
- Nandi, Asok Kumar; Siddhartha Chaudhuri; Sunil Kumar Mazumdar; Saktiprosad Ghosh (1984). "Effect of chlorine substitution on the structure and activity of 4-phenylthiosemicarbazide: crystal and molecular structure of 4-(4-chlorophenyl)thiosemicarbazide". J. Chem. Soc., Perkin Trans. 2 (11): 1729–1733. doi:10.1039/P29840001729.
External links
This article about an organic compound is a stub. You can help Misplaced Pages by expanding it. |