| Revision as of 22:30, 6 September 2022 editZefr (talk | contribs)Extended confirmed users, Pending changes reviewers69,475 edits move overly-technical images to gallery, WP:NOTTEXTBOOK; make unbiased case to include conjecture on talk pageTag: Reverted← Previous edit | Latest revision as of 20:10, 13 May 2024 edit undoZefr (talk | contribs)Extended confirmed users, Pending changes reviewers69,475 edits Reverted good faith edits by Ilonumbrel (talk): Clear enough in the existing articleTags: Twinkle Undo | ||
| (22 intermediate revisions by 12 users not shown) | |||
| Line 1: | Line 1: | ||
| {{Short description|Category of |
{{Short description|Category of polyphenol compound}} | ||
| {{distinguish|Flavonol}} | {{distinguish|Flavonol}} | ||
| {{use American English|date=September 2022}} | |||
| ] | ] | ||
| '''Flavan-3-ols''' (sometimes referred to as '''flavanols''') are a subgroup of ]s. They are derivatives of ]s that possess a 2-phenyl-3,4-dihydro-2''H''-chromen-3-ol skeleton. Flavan-3-ols are structurally diverse and include a range of compounds, such as ], ], ], ], ]s, ]s, ]s. They |
'''Flavan-3-ols''' (sometimes referred to as '''flavanols''') are a subgroup of ]s. They are derivatives of ]s that possess a 2-phenyl-3,4-dihydro-2''H''-chromen-3-ol skeleton. Flavan-3-ols are structurally diverse and include a range of compounds, such as ], ], ], ], ]s, ]s, ]s. They play a part in plant defense and are present in the majority of plants.<ref>{{cite journal | vauthors = Ullah C, Unsicker SB, Fellenberg C, Constabel CP, Schmidt A, Gershenzon J, Hammerbacher A | title = Flavan-3-ols Are an Effective Chemical Defense against Rust Infection | journal = Plant Physiology | volume = 175 | issue = 4 | pages = 1560–1578 | date = December 2017 | pmid = 29070515 | pmc = 5717727 | doi = 10.1104/pp.17.00842 }}</ref> | ||
| == Chemical structure == | == Chemical structure == | ||
| Line 8: | Line 10: | ||
| The single-molecule (monomer) catechin, or isomer epicatechin (see diagram), adds four hydroxyls to flavan-3-ol, making building blocks for concatenated polymers (]s) and higher order polymers (]s).<ref name="ReferenceA">{{cite book |title=OPC in Practice |vauthors=Schwitters B, Masquelier J |date=1995 |edition=3rd |oclc=45289285}}</ref> | The single-molecule (monomer) catechin, or isomer epicatechin (see diagram), adds four hydroxyls to flavan-3-ol, making building blocks for concatenated polymers (]s) and higher order polymers (]s).<ref name="ReferenceA">{{cite book |title=OPC in Practice |vauthors=Schwitters B, Masquelier J |date=1995 |edition=3rd |oclc=45289285}}</ref> | ||
| Flavan-3-ols possess two chiral carbons, meaning four ]s occur for each of them. They are distinguished from the yellow, ketone-containing flavonoids such as ] and ], which are called ]. Early use of the term '''bioflavonoid''' was imprecisely applied to include the flavanols, which are distinguished by absence of ketone(s). Catechin monomers, dimers, and trimers (oligomers) are colorless. |
Flavan-3-ols possess two chiral carbons, meaning four ]s occur for each of them. They are distinguished from the yellow, ketone-containing flavonoids such as ] and ], which are called ]. Early use of the term '''bioflavonoid''' was imprecisely applied to include the flavanols, which are distinguished by absence of ketone(s). Catechin monomers, dimers, and trimers (oligomers) are colorless. Higher order polymers, anthocyanidins, exhibit deepening reds and become ]s.<ref name="ReferenceA" /> | ||
| ] and ] are ]s, with (–)-epicatechin and (+)-catechin being the most common optical ]s found in nature. Catechin was first isolated from the plant extract ], from which it derives its name. Heating catechin past its point of decomposition releases ] (also called catechol), which explains the common origin of the names of these compounds. | ] and ] are ]s, with (–)-epicatechin and (+)-catechin being the most common optical ]s found in nature. Catechin was first isolated from the plant extract ], from which it derives its name. Heating catechin past its point of decomposition releases ] (also called catechol), which explains the common origin of the names of these compounds. | ||
| ] and ] contain an additional phenolic ] when compared to epicatechin and catechin, respectively, similar to the difference in ] compared to pyrocatechol. | ] and ] contain an additional phenolic ] when compared to ] and ], respectively, similar to the difference in ] compared to ]. | ||
| Catechin gallates are ] ]s of the catechins; an example is ], which is commonly the most abundant catechin in tea. ]s and ]s are oligomeric flavan-3-ols. | Catechin gallates are ] ]s of the catechins; an example is ], which is commonly the most abundant catechin in tea. ]s and ]s are oligomeric flavan-3-ols. | ||
| Line 21: | Line 23: | ||
| === Biosynthesis of (–)-epicatechin === | === Biosynthesis of (–)-epicatechin === | ||
| The flavonoids are products from a cinnamoyl-CoA starter unit, with chain extension using three molecules of malonyl-CoA. Reactions are catalyzed by a type III PKS enzyme. These enzyme do not use ACPSs, but instead employ coenzyme A esters and have a single active site to perform the necessary series of reactions, e.g. chain extension, condensation, and cyclization. Chain extension of 4-hydroxycinnamoyl-CoA with three molecules of malonyl-CoA gives initially a polyketide (Figure 1), which can be folded. These allow Claisen-like reactions to occur, generating aromatic rings.<ref name="Dewick2009p168" /><ref name="Winkel-Shirley2001p485-493" /> ] (FLIM) can be used to detect flavanols in plant cells.<ref>{{cite journal | vauthors = Mueller-Harvey I, Feucht W, Polster J, Trnková L, Burgos P, Parker AW, Botchway SW | title = Two-photon excitation with pico-second fluorescence lifetime imaging to detect nuclear association of flavanols | journal = Analytica Chimica Acta | volume = 719 | pages = 68–75 | date = March 2012 | pmid = 22340533 | doi = 10.1016/j.aca.2011.12.068 | s2cid = 24094780 | url = https://zenodo.org/record/1038611 }}</ref> | The flavonoids are products from a cinnamoyl-CoA starter unit, with chain extension using three molecules of malonyl-CoA. Reactions are catalyzed by a type III PKS enzyme. These enzyme do not use ACPSs, but instead employ coenzyme A esters and have a single active site to perform the necessary series of reactions, e.g. chain extension, condensation, and cyclization. Chain extension of 4-hydroxycinnamoyl-CoA with three molecules of malonyl-CoA gives initially a polyketide (Figure 1), which can be folded. These allow Claisen-like reactions to occur, generating aromatic rings.<ref name="Dewick2009p168">{{cite book | vauthors = Dewick PM | title = Medicinal Natural Products: a biosynthetic approach. | publisher = John Wiley & Sons | date = 2009 | page = 168 | isbn = 978-0-471-49641-0 }}</ref><ref name="Winkel-Shirley2001p485-493">{{cite journal | vauthors = Winkel-Shirley B | title = Flavonoid biosynthesis. A colorful model for genetics, biochemistry, cell biology, and biotechnology | journal = Plant Physiology | volume = 126 | issue = 2 | pages = 485–493 | date = June 2001 | pmid = 11402179 | pmc = 1540115 | doi = 10.1104/pp.126.2.485 }}</ref> ] (FLIM) can be used to detect flavanols in plant cells.<ref>{{cite journal | vauthors = Mueller-Harvey I, Feucht W, Polster J, Trnková L, Burgos P, Parker AW, Botchway SW | title = Two-photon excitation with pico-second fluorescence lifetime imaging to detect nuclear association of flavanols | journal = Analytica Chimica Acta | volume = 719 | pages = 68–75 | date = March 2012 | pmid = 22340533 | doi = 10.1016/j.aca.2011.12.068 | s2cid = 24094780 | url = https://zenodo.org/record/1038611 }}</ref> | ||
| ] | ] | ||
| '''Figure 1''':Schematic overview of the flavan-3-ol (–)-epicatechin biosynthesis in plants: Enzymes are indicated in blue, abbreviated as follows: E1, ] (PAL), E2, ] (TAL), E3, ], E4, ], E5, chalcone synthase (]), E6, ], E7, ], E8, ], E9, ], E10, anthocyanidin synthase (]), E11, ]. HSCoA, Coenzyme A. L-Tyr, L-tyrosine, L-Phe, L-phenylalanine. | '''Figure 1''':Schematic overview of the flavan-3-ol (–)-epicatechin biosynthesis in plants: Enzymes are indicated in blue, abbreviated as follows: E1, ] (PAL), E2, ] (TAL), E3, ], E4, ], E5, chalcone synthase (]), E6, ], E7, ], E8, ], E9, ], E10, anthocyanidin synthase (]), E11, ]. HSCoA, Coenzyme A. <small>L</small>-Tyr, <small>L</small>-tyrosine, <small>L</small>-Phe, <small>L</small>-phenylalanine. | ||
| === Aglycones === | === Aglycones === | ||
| Line 32: | Line 34: | ||
| ! Image!!Name!!Formula!!Oligomers | ! Image!!Name!!Formula!!Oligomers | ||
| |- | |- | ||
| | ] || ], C, (+)-Catechin || C<sub>15</sub>H<sub>14</sub>O<sub>6</sub> || ]s | | ] || ], C, (+)-Catechin || C<sub>15</sub>H<sub>14</sub>O<sub>6</sub> || ]s | ||
| |- | |- | ||
| | ] || ], EC, (–)-Epicatechin (cis) || C<sub>15</sub>H<sub>14</sub>O<sub>6</sub> || ]s | | ] || ], EC, (–)-Epicatechin (''cis'') || C<sub>15</sub>H<sub>14</sub>O<sub>6</sub> || ]s | ||
| |- | |- | ||
| | ] || ], EGC || C<sub>15</sub>H<sub>14</sub>O<sub>7</sub> || ]s | | ] || ], EGC || C<sub>15</sub>H<sub>14</sub>O<sub>7</sub> || ]s | ||
| Line 56: | Line 58: | ||
| {{see also|Polyphenols in tea|Polyphenols in wine|Cocoa bean#Phytochemicals and research}} | {{see also|Polyphenols in tea|Polyphenols in wine|Cocoa bean#Phytochemicals and research}} | ||
| ] | ] | ||
| Flavan-3-ols are abundant in ]s derived from the tea plant '']'', as well as in some ] (made from the seeds of '']''), although the content is affected considerably by processing, especially in ].<ref name="Hammerstone_2000">{{cite journal | vauthors = Hammerstone JF, Lazarus SA, Schmitz HH | title = Procyanidin content and variation in some commonly consumed foods | journal = The Journal of Nutrition | volume = 130 | issue = 8S Suppl | pages = 2086S–2092S | date = August 2000 | pmid = 10917927 | doi = 10.1093/jn/130.8.2086S | doi-access = free }}</ref><ref name="Payne_2010">{{cite journal | vauthors = Payne MJ, Hurst WJ, Miller KB, Rank C, Stuart DA | title = Impact of fermentation, drying, roasting, and Dutch processing on epicatechin and catechin content of cacao beans and cocoa ingredients | journal = Journal of Agricultural and Food Chemistry | volume = 58 | issue = 19 | pages = 10518–10527 | date = October 2010 | pmid = 20843086 | doi = 10.1021/jf102391q }}</ref> Flavan-3-ols are also present in the human diet in fruits, in particular ], ], ], and ].<ref>{{cite book | vauthors = Mabrym H, Harborne JB, Mabry TJ |title=The Flavonoids |publisher=Chapman and Hall |location=London |year=1975 |isbn=978-0-412-11960-6 }}</ref> Their content in food is variable and affected by various factors, such as ], ], and ].<ref name=":2">{{cite journal | vauthors = Manach C, Scalbert A, Morand C, Rémésy C, Jiménez L | title = Polyphenols: food sources and bioavailability | journal = The American Journal of Clinical Nutrition | volume = 79 | issue = 5 | pages = 727–747 | date = May 2004 | pmid = 15113710 | doi = 10.1093/ajcn/79.5.727 }}</ref> | Flavan-3-ols are abundant in ]s derived from the tea plant '']'', as well as in some ] (made from the seeds of '']''), although the content is affected considerably by processing, especially in ].<ref name="Hammerstone_2000">{{cite journal | vauthors = Hammerstone JF, Lazarus SA, Schmitz HH | title = Procyanidin content and variation in some commonly consumed foods | journal = The Journal of Nutrition | volume = 130 | issue = 8S Suppl | pages = 2086S–2092S | date = August 2000 | pmid = 10917927 | doi = 10.1093/jn/130.8.2086S | doi-access = free }}</ref><ref name="Payne_2010">{{cite journal | vauthors = Payne MJ, Hurst WJ, Miller KB, Rank C, Stuart DA | title = Impact of fermentation, drying, roasting, and Dutch processing on epicatechin and catechin content of cacao beans and cocoa ingredients | journal = Journal of Agricultural and Food Chemistry | volume = 58 | issue = 19 | pages = 10518–10527 | date = October 2010 | pmid = 20843086 | doi = 10.1021/jf102391q }}</ref> Flavan-3-ols are also present in the human diet in fruits, in particular ], ], ]s, and ].<ref>{{cite book | vauthors = Mabrym H, Harborne JB, Mabry TJ |title=The Flavonoids |publisher=Chapman and Hall |location=London |year=1975 |isbn=978-0-412-11960-6 }}</ref> Their content in food is variable and affected by various factors, such as ], ], and ].<ref name=":2">{{cite journal | vauthors = Manach C, Scalbert A, Morand C, Rémésy C, Jiménez L | title = Polyphenols: food sources and bioavailability | journal = The American Journal of Clinical Nutrition | volume = 79 | issue = 5 | pages = 727–747 | date = May 2004 | pmid = 15113710 | doi = 10.1093/ajcn/79.5.727 | doi-access = free }}</ref> | ||
| == Bioavailability and metabolism== | == Bioavailability and metabolism== | ||
| The ] of flavan-3-ols depends on the ], type of compound and their ] configuration.<ref name=":1">{{cite journal | vauthors = Del |
The ] of flavan-3-ols depends on the ], type of compound and their ] configuration.<ref name=":1">{{cite journal | vauthors = Del Río D, Rodríguez Mateos A, Spencer JP, Tognolini M, Borges G, Crozier A | title = Dietary (poly)phenolics in human health: structures, bioavailability, and evidence of protective effects against chronic diseases | journal = Antioxidants & Redox Signaling | volume = 18 | issue = 14 | pages = 1818–1892 | date = May 2013 | pmid = 22794138 | doi = 10.1089/ars.2012.4581 | pmc = 3619154 }}</ref> While monomeric flavan-3-ols are readily taken up, oligomeric forms are not absorbed.<ref name=":1" /><ref>{{cite journal | vauthors = Rodríguez Mateos A, Weber T, Skene SS, Ottaviani JI, Crozier A, Kelm M, Schroeter H, Heiss C | display-authors = 6 | title = Assessing the respective contributions of dietary flavanol monomers and procyanidins in mediating cardiovascular effects in humans: randomized, controlled, double-masked intervention trial | journal = The American Journal of Clinical Nutrition | volume = 108 | issue = 6 | pages = 1229–1237 | date = December 2018 | pmid = 30358831 | pmc = 6290365 | doi = 10.1093/ajcn/nqy229 }}</ref> Most data for human metabolism of flavan-3-ols are available for monomeric compounds, especially ]. These compounds are taken up and metabolized upon uptake in the ],<ref>{{cite journal | vauthors = Actis-Goretta L, Lévèques A, Rein M, Teml A, Schäfer C, Hofmann U, Li H, Schwab M, Eichelbaum M, Williamson G | display-authors = 6 | title = Intestinal absorption, metabolism, and excretion of (−)-epicatechin in healthy humans assessed by using an intestinal perfusion technique | journal = The American Journal of Clinical Nutrition | volume = 98 | issue = 4 | pages = 924–933 | date = October 2013 | pmid = 23864538 | doi = 10.3945/ajcn.113.065789 | doi-access = free }}</ref> mainly by ''O''-methylation and glucuronidation,<ref>{{cite journal | vauthors = Kuhnle G, Spencer JP, Schroeter H, Shenoy B, Debnam ES, Srai SK, Rice-Evans C, Hahn U | display-authors = 6 | title = Epicatechin and catechin are ''O''-methylated and glucuronidated in the small intestine | journal = Biochemical and Biophysical Research Communications | volume = 277 | issue = 2 | pages = 507–512 | date = October 2000 | pmid = 11032751 | doi = 10.1006/bbrc.2000.3701 }}</ref> and then further ] by the ]. The colonic ] has also an important role in the metabolism of flavan-3-ols and they are catabolized to smaller compounds such as 5-(3′/4′-dihydroxyphenyl)-γ-valerolactones and ].<ref>{{cite journal | vauthors = Das NP | title = Studies on flavonoid metabolism. Absorption and metabolism of (+)-catechin in man | journal = Biochemical Pharmacology | volume = 20 | issue = 12 | pages = 3435–3445 | date = December 1971 | pmid = 5132890 | doi = 10.1016/0006-2952(71)90449-7 }}</ref><ref name=":0" /> Only flavan-3-ols with an intact (epi)catechin moiety can be metabolized into 5-(3′/4′-dihydroxyphenyl)-γ-valerolactones (image in Gallery).<ref name="Ottaviani 9859"/> | ||
| ==Possible adverse effects== | ==Possible adverse effects== | ||
| Line 67: | Line 69: | ||
| ==Research== | ==Research== | ||
| {{See also|Cocoa bean#Phytochemicals and research}} | {{See also|Cocoa bean#Phytochemicals and research}} | ||
| Research has shown that flavan-3-ols may affect ], ], and ], with only minor effects demonstrated, as of 2019.<ref>{{cite journal | vauthors = Ried K, Fakler P, Stocks NP | title = Effect of cocoa on blood pressure | journal = The Cochrane Database of Systematic Reviews | volume = 4 | issue = 5 | pages = CD008893 | date = April 2017 | pmid = 28439881 | pmc = 6478304 | doi = 10.1002/14651858.CD008893.pub3 | collaboration = Cochrane Hypertension Group }}</ref><ref name="raman">{{cite journal | vauthors = Raman G, Avendano EE, Chen S, Wang J, Matson J, Gayer B, Novotny JA, Cassidy A | display-authors = 6 | title = Dietary intakes of flavan-3-ols and cardiometabolic health: systematic review and meta-analysis of randomized trials and prospective cohort studies | journal = The American Journal of Clinical Nutrition | volume = 110 | issue = 5 | pages = 1067–1078 | date = November 2019 | pmid = 31504087 | pmc = 6821550 | doi = 10.1093/ajcn/nqz178 }}</ref> | Research has shown that flavan-3-ols may affect ], ], and ], with only minor effects demonstrated, as of 2019.<ref>{{cite journal | vauthors = Ried K, Fakler P, Stocks NP | title = Effect of cocoa on blood pressure | journal = The Cochrane Database of Systematic Reviews | volume = 4 | issue = 5 | pages = CD008893 | date = April 2017 | pmid = 28439881 | pmc = 6478304 | doi = 10.1002/14651858.CD008893.pub3 | collaboration = Cochrane Hypertension Group }}</ref><ref name="raman">{{cite journal | vauthors = Raman G, Avendano EE, Chen S, Wang J, Matson J, Gayer B, Novotny JA, Cassidy A | display-authors = 6 | title = Dietary intakes of flavan-3-ols and cardiometabolic health: systematic review and meta-analysis of randomized trials and prospective cohort studies | journal = The American Journal of Clinical Nutrition | volume = 110 | issue = 5 | pages = 1067–1078 | date = November 2019 | pmid = 31504087 | pmc = 6821550 | doi = 10.1093/ajcn/nqz178 }}</ref> In 2015, the ] approved a ] for ]s containing 200 mg of flavanols, stating that such intake "may contribute to maintenance of vascular elasticity and normal blood flow".<ref name="EC-cocoa">{{cite web |title=Article 13 (5): Cocoa flavanols; Search filters: Claim status - authorised; search - flavanols |url=https://ec.europa.eu/food/safety/labelling_nutrition/claims/register/public/?event=search |publisher=European Commission, EU Register |access-date=8 September 2022 |date=31 March 2015}}</ref><ref name="efsa2014">{{Cite journal|title=Scientific Opinion on the modification of the authorisation of a health claim related to cocoa flavanols and maintenance of normal endothelium-dependent vasodilation pursuant to Article 13(5) of Regulation (EC) No 1924/20061 following a request in accordance with Article 19 of Regulation (EC) No 1924/2006|journal = EFSA Journal|volume = 12|issue = 5|date=2014|doi=10.2903/j.efsa.2014.3654|doi-access=free}}</ref> As of 2022, food-based evidence indicates that intake of 400–600 mg per day of flavan-3-ols could have a small positive effect on cardiovascular ]s.<ref>{{cite journal |last1=Crowe-White |first1=Kristi M |last2=Evans |first2=Levi W |last3=Kuhnle |first3=Gunter G C |last4=Milenkovic |first4=Dragan |last5=Stote |first5=Kim |last6=Wallace |first6=Taylor |last7=Handu |first7=Deepa |last8=Senkus |first8=Katelyn E |title=Flavan-3-ols and cardiometabolic health: First ever dietary bioactive guideline |journal=Advances in Nutrition |date=3 October 2022 |volume=13 |issue=6 |pages=2070–83 |doi=10.1093/advances/nmac105|pmid=36190328 |pmc=9776652 | url=https://academic.oup.com/advances/article/13/6/2070/6747118|doi-access=free }}</ref> | ||
| In 2014, the ] (EFSA) approved the following health claim for cocoa products containing 200 mg of flavanols and meeting the qualification in dietary supplement products: "cocoa flavanols help maintain the elasticity of blood vessels, which contributes to normal blood flow".<ref name="efsa2014">{{Cite journal|title=Scientific Opinion on the modification of the authorisation of a health claim related to cocoa flavanols and maintenance of normal endothelium-dependent vasodilation pursuant to Article 13(5) of Regulation (EC) No 1924/20061 following a request in accordance with Article 19 of Regulation (EC) No 1924/2006|journal = EFSA Journal|volume = 12|issue = 5|date=2014|doi=10.2903/j.efsa.2014.3654|doi-access=free}}</ref> | |||
| ==Gallery== | ==Gallery== | ||
| <gallery> | |||
| (-)-epicatechin in humans: implications for the assessment of efficacy, safety, and mechanisms of action of polyphenolic bioactives | language = En | journal = Scientific Reports | volume = 6 | issue = 1 | pages = 29034 | date = July 2016 | pmid = 27363516 | pmc = 4929566 | doi = 10.1038/srep29034 | bibcode = 2016NatSR...629034O }}</ref>]] | |||
| File:Schematic representation of (−)-epicatechin metabolism in humans as a function of time post-oral intake.jpg|Schematic representation of the flavan-3-ol (−)-epicatechin metabolism in humans as a function of time post-oral intake. SREM: structurally related (−)-epicatechin metabolites. 5C-RFM: 5-carbon ring fission metabolites. 3/1C-RFM: 3- and 1-carbon-side chain ring fission metabolites. The structures of the most abundant (−)-epicatechin metabolites present in the systemic circulation and in urine are depicted.<ref name=":0">{{cite journal | vauthors = Ottaviani JI, Borges G, Momma TY, Spencer JP, Keen CL, Crozier A, Schroeter H|display-authors=3 | title = The metabolome of (−)-epicatechin in humans: implications for the assessment of efficacy, safety, and mechanisms of action of polyphenolic bioactives | language = En | journal = Scientific Reports | volume = 6 | issue = 1 | pages = 29034 | date = July 2016 | pmid = 27363516 | pmc = 4929566 | doi = 10.1038/srep29034 | bibcode = 2016NatSR...629034O }}</ref> | |||
| File:Flavan-3-ol precursors of the microbial metabolite 5-(3′-4′-dihydroxyphenyl)-γ-valerolactone.jpg|Flavan-3-ol precursors of the microbial metabolite 5-(3′/4′-dihydroxyphenyl)-γ-valerolactone (gVL). Only compounds with intact (epi)catechin moiety result in the formation of γVL by the intestinal microbiome. ECG, (−)-epicatechin-3-''O''-gallate; EGCG, ]; EGC, ].<ref name="Ottaviani 9859">{{cite journal | vauthors = Ottaviani JI, Fong R, Kimball J, Ensunsa JL, Britten A, Lucarelli D, Luben R, Grace PB, Mawson DH, Tym A, Wierzbicki A, Khaw KT, Schroeter H, Kuhnle GG | display-authors = 6 | title = Evaluation at scale of microbiome-derived metabolites as biomarker of flavan-3-ol intake in epidemiological studies | language = En | journal = Scientific Reports | volume = 8 | issue = 1 | pages = 9859 | date = June 2018 | pmid = 29959422 | pmc = 6026136 | doi = 10.1038/s41598-018-28333-w | bibcode = 2018NatSR...8.9859O }}</ref> | |||
| {{clear}} | |||
| </gallery> | |||
| == References == | == References == | ||
| {{Reflist |
{{Reflist}} | ||
| <ref name="Dewick2009p168">{{cite book | vauthors = Dewick PM | title = Medicinal Natural Products: a biosynthetic approach. | publisher = John Wiley & Sons | date = 2009 | page = 168 | isbn = 978-0-471-49641-0 }}</ref><ref name="Winkel-Shirley2001p485-493">{{cite journal | vauthors = Winkel-Shirley B | title = Flavonoid biosynthesis. A colorful model for genetics, biochemistry, cell biology, and biotechnology | journal = Plant Physiology | volume = 126 | issue = 2 | pages = 485–493 | date = June 2001 | pmid = 11402179 | pmc = 1540115 | doi = 10.1104/pp.126.2.485 }}</ref> | |||
| }} | |||
| == External links == | == External links == | ||
| *{{Commons category-inline}} | *{{Commons category-inline}} | ||
| {{flavonoids}} | {{flavonoids}} | ||
| {{flavanol}} | {{flavanol}} | ||
| {{Chocolate}} | |||
| {{DEFAULTSORT:Flavan-3-Ol}} | {{DEFAULTSORT:Flavan-3-Ol}} | ||
Latest revision as of 20:10, 13 May 2024
Category of polyphenol compound Not to be confused with Flavonol.
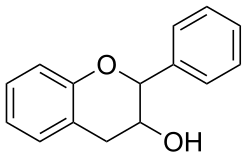
Flavan-3-ols (sometimes referred to as flavanols) are a subgroup of flavonoids. They are derivatives of flavans that possess a 2-phenyl-3,4-dihydro-2H-chromen-3-ol skeleton. Flavan-3-ols are structurally diverse and include a range of compounds, such as catechin, epicatechin gallate, epigallocatechin, epigallocatechin gallate, proanthocyanidins, theaflavins, thearubigins. They play a part in plant defense and are present in the majority of plants.
Chemical structure
The single-molecule (monomer) catechin, or isomer epicatechin (see diagram), adds four hydroxyls to flavan-3-ol, making building blocks for concatenated polymers (proanthocyanidins) and higher order polymers (anthocyanidins).
Flavan-3-ols possess two chiral carbons, meaning four diastereoisomers occur for each of them. They are distinguished from the yellow, ketone-containing flavonoids such as quercitin and rutin, which are called flavonols. Early use of the term bioflavonoid was imprecisely applied to include the flavanols, which are distinguished by absence of ketone(s). Catechin monomers, dimers, and trimers (oligomers) are colorless. Higher order polymers, anthocyanidins, exhibit deepening reds and become tannins.
Catechin and epicatechin are epimers, with (–)-epicatechin and (+)-catechin being the most common optical isomers found in nature. Catechin was first isolated from the plant extract catechu, from which it derives its name. Heating catechin past its point of decomposition releases pyrocatechol (also called catechol), which explains the common origin of the names of these compounds.
Epigallocatechin and gallocatechin contain an additional phenolic hydroxyl group when compared to epicatechin and catechin, respectively, similar to the difference in pyrogallol compared to pyrocatechol.
Catechin gallates are gallic acid esters of the catechins; an example is epigallocatechin gallate, which is commonly the most abundant catechin in tea. Proanthocyanidins and thearubigins are oligomeric flavan-3-ols.
In contrast to many other flavonoids, flavan-3-ols do not generally exist as glycosides in plants.
Biosynthesis of (–)-epicatechin
The flavonoids are products from a cinnamoyl-CoA starter unit, with chain extension using three molecules of malonyl-CoA. Reactions are catalyzed by a type III PKS enzyme. These enzyme do not use ACPSs, but instead employ coenzyme A esters and have a single active site to perform the necessary series of reactions, e.g. chain extension, condensation, and cyclization. Chain extension of 4-hydroxycinnamoyl-CoA with three molecules of malonyl-CoA gives initially a polyketide (Figure 1), which can be folded. These allow Claisen-like reactions to occur, generating aromatic rings. Fluorescence-lifetime imaging microscopy (FLIM) can be used to detect flavanols in plant cells.
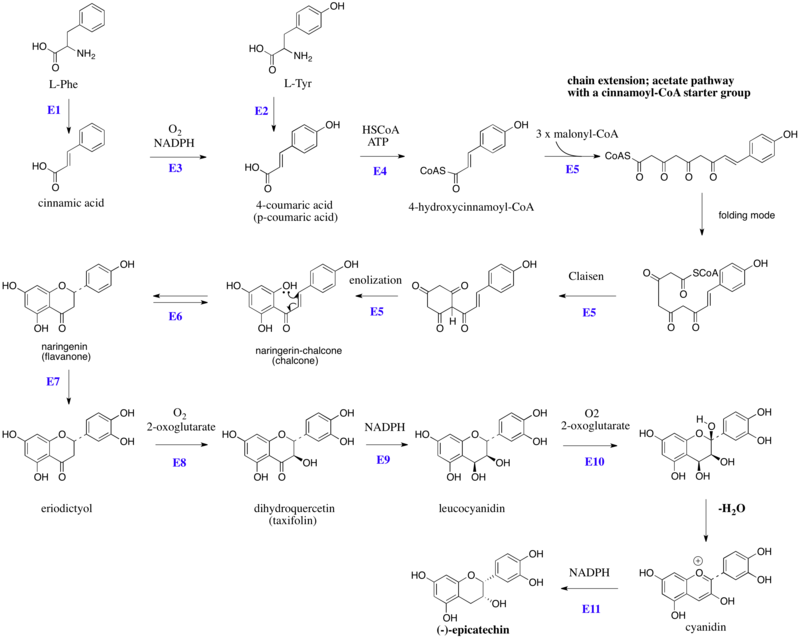
Figure 1:Schematic overview of the flavan-3-ol (–)-epicatechin biosynthesis in plants: Enzymes are indicated in blue, abbreviated as follows: E1, phenylalanine ammonia lyase (PAL), E2, tyrosine ammonia lyase (TAL), E3, cinnamate 4-hydroxylase, E4, 4-coumaroyl: CoA-ligase, E5, chalcone synthase (naringenin-chalcone synthase), E6, chalcone isomerase, E7, Flavonoid 3'-hydroxylase, E8, flavonone 3-hydroxylase, E9, dihydroflavanol 4-reductase, E10, anthocyanidin synthase (leucoanthocyanidin dioxygenase), E11, anthocyanidin reductase. HSCoA, Coenzyme A. L-Tyr, L-tyrosine, L-Phe, L-phenylalanine.
Aglycones
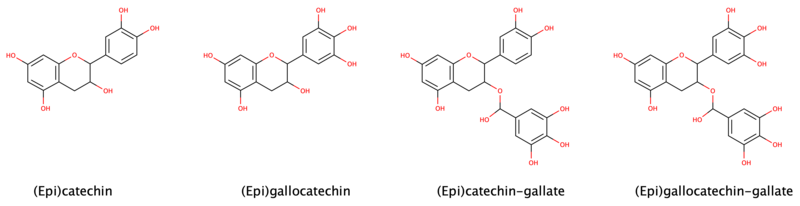
| Image | Name | Formula | Oligomers |
|---|---|---|---|
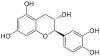 |
Catechin, C, (+)-Catechin | C15H14O6 | Procyanidins |
 |
Epicatechin, EC, (–)-Epicatechin (cis) | C15H14O6 | Procyanidins |
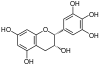 |
Epigallocatechin, EGC | C15H14O7 | Prodelphinidins |
 |
Epicatechin gallate, ECG | C22H18O10 | |
 |
Epigallocatechin gallate, EGCG, (–)-Epigallocatechin gallate |
C22H18O11 | |
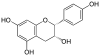 |
Epiafzelechin | C15H14O5 | |
| Fisetinidol | C15H14O5 | ||
| Guibourtinidol | C15H14O4 | Proguibourtinidins | |
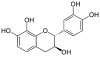 |
Mesquitol | C15H14O6 | |
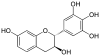 |
Robinetinidol | C15H14O6 | Prorobinetinidins |
Dietary sources
See also: Polyphenols in tea, Polyphenols in wine, and Cocoa bean § Phytochemicals and research
Flavan-3-ols are abundant in teas derived from the tea plant Camellia sinensis, as well as in some cocoas (made from the seeds of Theobroma cacao), although the content is affected considerably by processing, especially in chocolate. Flavan-3-ols are also present in the human diet in fruits, in particular pome fruits, berries, vegetables, and wine. Their content in food is variable and affected by various factors, such as cultivar, processing, and preparation.
Bioavailability and metabolism
The bioavailability of flavan-3-ols depends on the food matrix, type of compound and their stereochemical configuration. While monomeric flavan-3-ols are readily taken up, oligomeric forms are not absorbed. Most data for human metabolism of flavan-3-ols are available for monomeric compounds, especially epiatechin. These compounds are taken up and metabolized upon uptake in the jejunum, mainly by O-methylation and glucuronidation, and then further metabolized by the liver. The colonic microbiome has also an important role in the metabolism of flavan-3-ols and they are catabolized to smaller compounds such as 5-(3′/4′-dihydroxyphenyl)-γ-valerolactones and hippuric acid. Only flavan-3-ols with an intact (epi)catechin moiety can be metabolized into 5-(3′/4′-dihydroxyphenyl)-γ-valerolactones (image in Gallery).
Possible adverse effects
As catechins in green tea extract can be hepatotoxic, Health Canada and EFSA have advised for caution, recommending intake should not exceed 800 mg per day.
Research
See also: Cocoa bean § Phytochemicals and researchResearch has shown that flavan-3-ols may affect vascular function, blood pressure, and blood lipids, with only minor effects demonstrated, as of 2019. In 2015, the European Commission approved a health claim for cocoa solids containing 200 mg of flavanols, stating that such intake "may contribute to maintenance of vascular elasticity and normal blood flow". As of 2022, food-based evidence indicates that intake of 400–600 mg per day of flavan-3-ols could have a small positive effect on cardiovascular biomarkers.
Gallery
-
 Schematic representation of the flavan-3-ol (−)-epicatechin metabolism in humans as a function of time post-oral intake. SREM: structurally related (−)-epicatechin metabolites. 5C-RFM: 5-carbon ring fission metabolites. 3/1C-RFM: 3- and 1-carbon-side chain ring fission metabolites. The structures of the most abundant (−)-epicatechin metabolites present in the systemic circulation and in urine are depicted.
Schematic representation of the flavan-3-ol (−)-epicatechin metabolism in humans as a function of time post-oral intake. SREM: structurally related (−)-epicatechin metabolites. 5C-RFM: 5-carbon ring fission metabolites. 3/1C-RFM: 3- and 1-carbon-side chain ring fission metabolites. The structures of the most abundant (−)-epicatechin metabolites present in the systemic circulation and in urine are depicted.
-
 Flavan-3-ol precursors of the microbial metabolite 5-(3′/4′-dihydroxyphenyl)-γ-valerolactone (gVL). Only compounds with intact (epi)catechin moiety result in the formation of γVL by the intestinal microbiome. ECG, (−)-epicatechin-3-O-gallate; EGCG, Epigallocatechin gallate; EGC, Epigallocatechin.
Flavan-3-ol precursors of the microbial metabolite 5-(3′/4′-dihydroxyphenyl)-γ-valerolactone (gVL). Only compounds with intact (epi)catechin moiety result in the formation of γVL by the intestinal microbiome. ECG, (−)-epicatechin-3-O-gallate; EGCG, Epigallocatechin gallate; EGC, Epigallocatechin.
References
- Ullah C, Unsicker SB, Fellenberg C, Constabel CP, Schmidt A, Gershenzon J, Hammerbacher A (December 2017). "Flavan-3-ols Are an Effective Chemical Defense against Rust Infection". Plant Physiology. 175 (4): 1560–1578. doi:10.1104/pp.17.00842. PMC 5717727. PMID 29070515.
- ^ Schwitters B, Masquelier J (1995). OPC in Practice (3rd ed.). OCLC 45289285.
- Del Rio D, Rodriguez-Mateos A, Spencer JP, Tognolini M, Borges G, Crozier A (May 2013). "Dietary (poly)phenolics in human health: structures, bioavailability, and evidence of protective effects against chronic diseases". Antioxidants & Redox Signaling. 18 (14): 1818–1892. doi:10.1089/ars.2012.4581. PMC 3619154. PMID 22794138.
- Dewick PM (2009). Medicinal Natural Products: a biosynthetic approach. John Wiley & Sons. p. 168. ISBN 978-0-471-49641-0.
- Winkel-Shirley B (June 2001). "Flavonoid biosynthesis. A colorful model for genetics, biochemistry, cell biology, and biotechnology". Plant Physiology. 126 (2): 485–493. doi:10.1104/pp.126.2.485. PMC 1540115. PMID 11402179.
- Mueller-Harvey I, Feucht W, Polster J, Trnková L, Burgos P, Parker AW, Botchway SW (March 2012). "Two-photon excitation with pico-second fluorescence lifetime imaging to detect nuclear association of flavanols". Analytica Chimica Acta. 719: 68–75. doi:10.1016/j.aca.2011.12.068. PMID 22340533. S2CID 24094780.
- "Database on polyphenol content in foods, v. 3.6". Phenol Explorer. 2016.
- Hammerstone JF, Lazarus SA, Schmitz HH (August 2000). "Procyanidin content and variation in some commonly consumed foods". The Journal of Nutrition. 130 (8S Suppl): 2086S – 2092S. doi:10.1093/jn/130.8.2086S. PMID 10917927.
- Payne MJ, Hurst WJ, Miller KB, Rank C, Stuart DA (October 2010). "Impact of fermentation, drying, roasting, and Dutch processing on epicatechin and catechin content of cacao beans and cocoa ingredients". Journal of Agricultural and Food Chemistry. 58 (19): 10518–10527. doi:10.1021/jf102391q. PMID 20843086.
- Mabrym H, Harborne JB, Mabry TJ (1975). The Flavonoids. London: Chapman and Hall. ISBN 978-0-412-11960-6.
- Manach C, Scalbert A, Morand C, Rémésy C, Jiménez L (May 2004). "Polyphenols: food sources and bioavailability". The American Journal of Clinical Nutrition. 79 (5): 727–747. doi:10.1093/ajcn/79.5.727. PMID 15113710.
- ^ Del Río D, Rodríguez Mateos A, Spencer JP, Tognolini M, Borges G, Crozier A (May 2013). "Dietary (poly)phenolics in human health: structures, bioavailability, and evidence of protective effects against chronic diseases". Antioxidants & Redox Signaling. 18 (14): 1818–1892. doi:10.1089/ars.2012.4581. PMC 3619154. PMID 22794138.
- Rodríguez Mateos A, Weber T, Skene SS, Ottaviani JI, Crozier A, Kelm M, et al. (December 2018). "Assessing the respective contributions of dietary flavanol monomers and procyanidins in mediating cardiovascular effects in humans: randomized, controlled, double-masked intervention trial". The American Journal of Clinical Nutrition. 108 (6): 1229–1237. doi:10.1093/ajcn/nqy229. PMC 6290365. PMID 30358831.
- Actis-Goretta L, Lévèques A, Rein M, Teml A, Schäfer C, Hofmann U, et al. (October 2013). "Intestinal absorption, metabolism, and excretion of (−)-epicatechin in healthy humans assessed by using an intestinal perfusion technique". The American Journal of Clinical Nutrition. 98 (4): 924–933. doi:10.3945/ajcn.113.065789. PMID 23864538.
- Kuhnle G, Spencer JP, Schroeter H, Shenoy B, Debnam ES, Srai SK, et al. (October 2000). "Epicatechin and catechin are O-methylated and glucuronidated in the small intestine". Biochemical and Biophysical Research Communications. 277 (2): 507–512. doi:10.1006/bbrc.2000.3701. PMID 11032751.
- Das NP (December 1971). "Studies on flavonoid metabolism. Absorption and metabolism of (+)-catechin in man". Biochemical Pharmacology. 20 (12): 3435–3445. doi:10.1016/0006-2952(71)90449-7. PMID 5132890.
- ^ Ottaviani JI, Borges G, Momma TY, et al. (July 2016). "The metabolome of [2-C](−)-epicatechin in humans: implications for the assessment of efficacy, safety, and mechanisms of action of polyphenolic bioactives". Scientific Reports. 6 (1): 29034. Bibcode:2016NatSR...629034O. doi:10.1038/srep29034. PMC 4929566. PMID 27363516.
- ^ Ottaviani JI, Fong R, Kimball J, Ensunsa JL, Britten A, Lucarelli D, et al. (June 2018). "Evaluation at scale of microbiome-derived metabolites as biomarker of flavan-3-ol intake in epidemiological studies". Scientific Reports. 8 (1): 9859. Bibcode:2018NatSR...8.9859O. doi:10.1038/s41598-018-28333-w. PMC 6026136. PMID 29959422.
- Health Canada (12 December 2017). "Summary Safety Review - Green tea extract-containing natural health products - Assessing the potential risk of liver injury (hepatotoxicity)". Health Canada, Government of Canada. Retrieved 2022-05-06.
- Younes M, Aggett P, Aguilar F, Crebelli R, Dusemund B, Filipič M, et al. (April 2018). "Scientific opinion on the safety of green tea catechins". EFSA Journal. 16 (4): e05239. doi:10.2903/j.efsa.2018.5239. PMC 7009618. PMID 32625874.
- Ried K, Fakler P, Stocks NP, et al. (Cochrane Hypertension Group) (April 2017). "Effect of cocoa on blood pressure". The Cochrane Database of Systematic Reviews. 4 (5): CD008893. doi:10.1002/14651858.CD008893.pub3. PMC 6478304. PMID 28439881.
- Raman G, Avendano EE, Chen S, Wang J, Matson J, Gayer B, et al. (November 2019). "Dietary intakes of flavan-3-ols and cardiometabolic health: systematic review and meta-analysis of randomized trials and prospective cohort studies". The American Journal of Clinical Nutrition. 110 (5): 1067–1078. doi:10.1093/ajcn/nqz178. PMC 6821550. PMID 31504087.
- "Article 13 (5): Cocoa flavanols; Search filters: Claim status - authorised; search - flavanols". European Commission, EU Register. 31 March 2015. Retrieved 8 September 2022.
- "Scientific Opinion on the modification of the authorisation of a health claim related to cocoa flavanols and maintenance of normal endothelium-dependent vasodilation pursuant to Article 13(5) of Regulation (EC) No 1924/20061 following a request in accordance with Article 19 of Regulation (EC) No 1924/2006". EFSA Journal. 12 (5). 2014. doi:10.2903/j.efsa.2014.3654.
- Crowe-White, Kristi M; Evans, Levi W; Kuhnle, Gunter G C; Milenkovic, Dragan; Stote, Kim; Wallace, Taylor; Handu, Deepa; Senkus, Katelyn E (3 October 2022). "Flavan-3-ols and cardiometabolic health: First ever dietary bioactive guideline". Advances in Nutrition. 13 (6): 2070–83. doi:10.1093/advances/nmac105. PMC 9776652. PMID 36190328.
External links
 Media related to Flavan-3-ols at Wikimedia Commons
Media related to Flavan-3-ols at Wikimedia Commons
| Types of flavonoids | |||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Flavonoids |
| ||||||||||||||||||||||||||||||||||||||||
| Flavonoid biosynthesis | |||||||||||||||||||||||||||||||||||||||||
| Flavan-3-ols and their glycosides | |
|---|---|
| Flavan-3-ols | |
| O-methylated flavan-3ols |
|
| Glycosides |
|
| Acetylated | Phylloflavan |
| Gallate esters | |
| Misc. | |