| |
| Names | |
|---|---|
| IUPAC name N--L-glutamic acid | |
| Systematic IUPAC name (2S)-2-(5-Hydroxypyridine-3-carboxamido)pentanoic acid | |
| Other names Nooglutil | |
| Identifiers | |
| CAS Number | |
| 3D model (JSmol) | |
| ChemSpider | |
| PubChem CID | |
| UNII | |
| CompTox Dashboard (EPA) | |
InChI
| |
SMILES
| |
| Properties | |
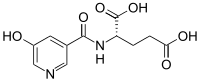
| Chemical formula | C11H12N2O6 |
| Molar mass | 268.225 g·mol |
| Except where otherwise noted, data are given for materials in their standard state (at 25 °C , 100 kPa). Infobox references | |
Nooglutyl is a nootropic agent that was studied at the Research Institute of Pharmacology, Russian Academy of Medical Sciences as a potential treatment for amnesia.
In animal models, it has a variety of central nervous system effects.
Nooglutyl has some structural similarities to picamilon (N-Nicotinoyl-GABA). Nooglutyl is N-5-HydroxyNicotinoyl-L-Glutamic acid.
See also
References
- Flekhter, Oxana B. (2000). "Nooglutil, Russian Academy of Medical Science". Current Opinion in Central & Peripheral Nervous System Investigational Drugs. 2 (4): 491–497.
- V. V. Yasnetsov; V. A. Pravdivtsev; V. M. Popov; T. A. Voronina; N. M. Kiseleva; S. B. Kozlov (May 1995). "Antimotion Effect of Nooglutyl and Its Neuronal Mechanism". Bulletin of Experimental Biology and Medicine. 119 (5): 515–516. doi:10.1007/BF02543440. PMID 7579248. S2CID 6344628.
- Voronina, TA; Borlikova, GG; Garibova, TL; Proskuryakova, TV; Petrichenko, OB; Burd, SG; Avakyan, GN (2002). "Effect of nooglutil on benzodiazepine withdrawal syndrome and binding of 3H-spiperone with D2 receptors in rat striatum". Bulletin of Experimental Biology and Medicine. 134 (5): 448–50. doi:10.1023/a:1022634112815. PMID 12802448. S2CID 142803.
- Garibova, TL; Galaeva, IP; Voronina, TA; Kraĭneva, VA; Kapitsa, IG; Kirichenko, SV; Makarenko, AN; Mirzoian, GR; Kuznetsova, EA (2003). "Effect of nooglutil on rats with intracerebral posttraumatic hematoma (hemorrhagic stroke)". Eksperimental'naia i Klinicheskaia Farmakologiia. 66 (3): 13–6. PMID 12924225.
- Povarova, OV; Garibova, TL; Kalenikova, EI; Galaeva, IP; Kraĭneva, VA; Medvedev, OS; Voronina, TA (2004). "Effect of phenyl-tert-butylnitrone, mexidol and nooglutil on the ischemic lesion zone and memory in rats following middle cerebral artery occlusion". Eksperimental'naia i Klinicheskaia Farmakologiia. 67 (1): 3–6. PMID 15079898.
This drug article relating to the nervous system is a stub. You can help Misplaced Pages by expanding it. |