| Revision as of 19:58, 1 September 2011 editBogBot (talk | contribs)Bots53,132 edits populated new fields in {{drugbox}} and reordered per bot approval. Report errors and suggestions to User_talk:BogBot← Previous edit | Latest revision as of 15:34, 8 October 2024 edit undoJWBE (talk | contribs)Extended confirmed users10,127 edits removed Category:Anilines; added Category:4-Aminophenyl compounds using HotCat | ||
| (25 intermediate revisions by 16 users not shown) | |||
| Line 1: | Line 1: | ||
| {{short description|Chemical used to treat pulmonary issues}} | |||
| {{Drugbox | {{Drugbox | ||
| | Watchedfields = changed | | Watchedfields = changed | ||
| | verifiedrevid = |
| verifiedrevid = 470472993 | ||
| | IUPAC_name = 4-Amino-''N''-(3-methoxypyrazinyl)benzenesulfonamide | | IUPAC_name = 4-Amino-''N''-(3-methoxypyrazinyl)benzenesulfonamide | ||
| | image = |
| image = Sulfalene2DCSD.svg | ||
| | width = | | width = | ||
| | alt = | | alt = | ||
| | image2 = | | image2 = | ||
| | width2 = | | width2 = | ||
| | drug_name = | |||
| | imagename = <!-- else may use drug_name --> | |||
| | drug_name = <!-- else may use imagename --> | |||
| | caption = | | caption = | ||
| <!--Clinical data--> | <!--Clinical data--> | ||
| | tradename = | | tradename = Eadazine, Kelfizina, Kelfizine W, Longum | ||
| | Drugs.com = {{drugs.com|international|sulfametopyrazine}} | | Drugs.com = {{drugs.com|international|sulfametopyrazine}} | ||
| | licence_EU = <!-- EMA requires brand name --> | | licence_EU = <!-- EMA requires brand name --> | ||
| Line 27: | Line 26: | ||
| | legal_status = | | legal_status = | ||
| | dependency_liability = | | dependency_liability = | ||
| | routes_of_administration = Oral<ref name="MIMS">{{cite web | url = http://www.cimsasia.com/USA/drug/info/sulfalene/?q |
| routes_of_administration = Oral<ref name="MIMS">{{cite web | url = http://www.cimsasia.com/USA/drug/info/sulfalene/?q=Sulphonamides&type=full | title = Sulfalene | work = MIMS Drug Information System | access-date = 26 August 2011}}</ref> | ||
| <!--Pharmacokinetic data--> | <!--Pharmacokinetic data--> | ||
| Line 37: | Line 36: | ||
| <!--Identifiers--> | <!--Identifiers--> | ||
| | CAS_number_Ref = {{cascite|correct|??}} | |||
| | CAS_number = 152-47-6 | | CAS_number = 152-47-6 | ||
| | CAS_supplemental = | | CAS_supplemental = | ||
| Line 42: | Line 42: | ||
| | ATC_prefix = J01 | | ATC_prefix = J01 | ||
| | ATC_suffix = ED02 | | ATC_suffix = ED02 | ||
| | ATC_supplemental = |
| ATC_supplemental = {{ATCvet|J01|EQ19}} | ||
| | PubChem = 9047 | | PubChem = 9047 | ||
| | PubChemSubstance = | | PubChemSubstance = | ||
| Line 56: | Line 56: | ||
| | ChEBI_Ref = {{ebicite|correct|EBI}} | | ChEBI_Ref = {{ebicite|correct|EBI}} | ||
| | ChEMBL_Ref = {{ebicite|correct|EBI}} | | ChEMBL_Ref = {{ebicite|correct|EBI}} | ||
| <!--Chemical data--> | <!--Chemical data--> | ||
| | C=11 | H=12 | N=4 | O=3 | S=1 |
| C=11 | H=12 | N=4 | O=3 | S=1 | ||
| ⚫ | | SMILES = O=S(=O)(Nc1nccnc1OC)c2ccc(N)cc2 | ||
| | molecular_weight = 280.304 g/mol | |||
| ⚫ | | |
||
| | InChI = 1/C11H12N4O3S/c1-18-11-10(13-6-7-14-11)15-19(16,17)9-4-2-8(12)3-5-9/h2-7H,12H2,1H3,(H,13,15) | |||
| | InChIKey = KXRZBTAEDBELFD-UHFFFAOYAH | |||
| | StdInChI_Ref = {{stdinchicite|correct|chemspider}} | | StdInChI_Ref = {{stdinchicite|correct|chemspider}} | ||
| | StdInChI = 1S/C11H12N4O3S/c1-18-11-10(13-6-7-14-11)15-19(16,17)9-4-2-8(12)3-5-9/h2-7H,12H2,1H3,(H,13,15) | | StdInChI = 1S/C11H12N4O3S/c1-18-11-10(13-6-7-14-11)15-19(16,17)9-4-2-8(12)3-5-9/h2-7H,12H2,1H3,(H,13,15) | ||
| Line 79: | Line 75: | ||
| }} | }} | ||
| '''Sulfalene''' is a ] ] used for the treatment of ], ]s and ].<ref name="DrugBank">{{DrugBank|DB00664}}</ref> | '''Sulfalene''' (], ]) or '''sulfametopyrazine''' (]) is a long-acting ] ] used for the treatment of ], ]s and ].<ref name="DrugBank">{{DrugBank|DB00664}}</ref><ref name = MD/> As of 2014 there were only two countries in which it is currently still marketed: ] and ].<ref name = MD>{{cite web|title=Sulfametopyrazine|work=Martindale: The Complete Drug Reference|publisher=Pharmaceutical Press|date=9 May 2013|access-date=28 March 2014| veditors = Brayfield A |url=http://www.medicinescomplete.com/mc/martindale/current/4920-k.htm}}</ref> | ||
| It was discovered by researchers at ] and first published in 1960 and was marketed as Kelfizina.<ref name="pmid5332105">{{cite journal | vauthors = Baruffa G | title = Clinical trials in Plasmodium falciparum malaria with a long-acting sulphonamide | journal = Transactions of the Royal Society of Tropical Medicine and Hygiene | volume = 60 | issue = 2 | pages = 222–4 | date = 1966 | pmid = 5332105 | doi = 10.1016/0035-9203(66)90030-7 }}</ref><ref>Per prior citation, the first publication: {{cite journal | vauthors = Camerino B, Palamidessi G | date = 1960 | title = Derivati della parazina II. Sulfonamdopir |language=it | journal = Gazz Chim Ital | volume = 90 | pages = 1802–1815 }}</ref> | |||
| ==See also== | |||
| *] | |||
| == References == | == References == | ||
| <references/> | <references/> | ||
| Line 86: | Line 86: | ||
| {{Sulfonamides and trimethoprim}} | {{Sulfonamides and trimethoprim}} | ||
| ] | ] | ||
| ] | ] | ||
| ] | ] | ||
| ] | ] | ||
| ] | |||
| ] | |||
| ] | |||
| {{antibiotic-stub}} | {{antibiotic-stub}} | ||
| ] | |||
Latest revision as of 15:34, 8 October 2024
Chemical used to treat pulmonary issues Pharmaceutical compound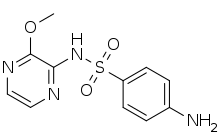 | |
| Clinical data | |
|---|---|
| Trade names | Eadazine, Kelfizina, Kelfizine W, Longum |
| Other names | Sulfametopyrazine |
| AHFS/Drugs.com | International Drug Names |
| Routes of administration | Oral |
| ATC code | |
| Pharmacokinetic data | |
| Protein binding | 60 to 80% |
| Elimination half-life | 60 to 65 hours |
| Excretion | Urinary |
| Identifiers | |
IUPAC name
| |
| CAS Number | |
| PubChem CID | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.005.278 |
| Chemical and physical data | |
| Formula | C11H12N4O3S |
| Molar mass | 280.30 g·mol |
| 3D model (JSmol) | |
SMILES
| |
InChI
| |
| (verify) | |
Sulfalene (INN, USAN) or sulfametopyrazine (BAN) is a long-acting sulfonamide antibacterial used for the treatment of chronic bronchitis, urinary tract infections and malaria. As of 2014 there were only two countries in which it is currently still marketed: Thailand and Ireland.
It was discovered by researchers at Farmitalia and first published in 1960 and was marketed as Kelfizina.
See also
References
- ^ "Sulfalene". MIMS Drug Information System. Retrieved 26 August 2011.
- DrugBank DB00664
- ^ Brayfield A, ed. (9 May 2013). "Sulfametopyrazine". Martindale: The Complete Drug Reference. Pharmaceutical Press. Retrieved 28 March 2014.
- Baruffa G (1966). "Clinical trials in Plasmodium falciparum malaria with a long-acting sulphonamide". Transactions of the Royal Society of Tropical Medicine and Hygiene. 60 (2): 222–4. doi:10.1016/0035-9203(66)90030-7. PMID 5332105.
- Per prior citation, the first publication: Camerino B, Palamidessi G (1960). "Derivati della parazina II. Sulfonamdopir". Gazz Chim Ital (in Italian). 90: 1802–1815.
| Antibacterials that inhibit nucleic acid (J01E, J01M) | |||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Antifolates (inhibit bacterial purine metabolism, thereby inhibiting DNA and RNA synthesis) |
| ||||||||||||||||
| Quinolones (inhibit bacterial topoisomerase and/or DNA gyrase, thereby inhibiting DNA replication) |
| ||||||||||||||||
| Anaerobic DNA inhibitors |
| ||||||||||||||||
| RNA synthesis |
| ||||||||||||||||
| |||||||||||||||||
This systemic antibiotic-related article is a stub. You can help Misplaced Pages by expanding it. |