This is an old revision of this page, as edited by Fmadd (talk | contribs) at 08:35, 22 April 2016. The present address (URL) is a permanent link to this revision, which may differ significantly from the current revision.
Revision as of 08:35, 22 April 2016 by Fmadd (talk | contribs)(diff) ← Previous revision | Latest revision (diff) | Newer revision → (diff) Pharmaceutical compound | |
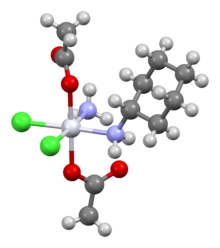 | |
| Clinical data | |
|---|---|
| Other names | BMY 45594 BMS 182751 (OC-6-43)-bis(acetato)amminedichlorocyclohexylamine platinum(IV) |
| Routes of administration | Oral |
| ATC code | |
| Identifiers | |
IUPAC name
| |
| CAS Number | |
| PubChem CID | |
| UNII | |
| ChEBI | |
| CompTox Dashboard (EPA) | |
| Chemical and physical data | |
| Formula | C10H22Cl2N2O4Pt |
| Molar mass | 500.277 g/mol g·mol |
| (verify) | |
Satraplatin (INN, codenamed JM216) is a platinum-based antineoplastic agent that is under investigation as one treatment of patients with advanced prostate cancer who have failed previous chemotherapy. It has not yet received approval from the U.S. Food and Drug Administration. First mentioned in the medical literature in 1993, satraplatin is the first orally active platinum-based chemotherapeutic drug; other available platinum analogues—cisplatin, carboplatin, and oxaliplatin—must be given intravenously.
It is made available in the United States jointly by Spectrum Pharmaceuticals and GPC Biotech under the name SPERA (SatraPlatin Expanded Rapid Access).
The drug has also been used in the treatment of lung and ovarian cancers. The proposed mode of action is that the compound binds to the DNA of cancer cells rendering them incapable of dividing.
References
- Wheate, Nial J.; Walker, Shonagh; Craig, Gemma E.; Oun, Rabbab (2010). "The status of platinum anticancer drugs in the clinic and in clinical trials". Dalton Transactions. 39 (35): 8113–27. doi:10.1039/C0DT00292E. PMID 20593091.
- Kelland LR, Abel G, McKeage MJ, et al. (1993). "Preclinical antitumor evaluation of bis-acetato-ammine-dichloro-cyclohexylamine platinum(IV): an [[orally active]] platinum drug" (PDF). Cancer Res. 53 (11): 2581–6. PMID 8388318.
{{cite journal}}: URL–wikilink conflict (help) - Choy H, Park C, Yao M (2008). "Current status and future prospects for satraplatin, an oral platinum analogue". Clin Cancer Res. 14 (6): 1633–8. doi:10.1158/1078-0432.CCR-07-2176. PMID 18347164.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - Satraplatin — Spectrum Pharmaceuticals
| Platinum compounds | |||
|---|---|---|---|
| Pt(−II) | |||
| Pt(0) | |||
| Pt(II) |
| ||
| Pt(IV) | |||
| Pt(V) | |||
| Pt(VI) | |||
This antineoplastic or immunomodulatory drug article is a stub. You can help Misplaced Pages by expanding it. |