| |
| |
 Solution of CsAu(left), pure CsAu(right) | |
| Names | |
|---|---|
| IUPAC name Caesium auride | |
| Identifiers | |
| CAS Number | |
| 3D model (JSmol) |
|
| ChemSpider | |
| PubChem CID | |
InChI
| |
SMILES
| |
| Properties | |
| Chemical formula | AuCs |
| Molar mass | 329.872022 g·mol |
| Appearance | Yellow crystals |
| Melting point | 580 °C (1,076 °F; 853 K) |
| Solubility in water | reacts violently |
| Structure | |
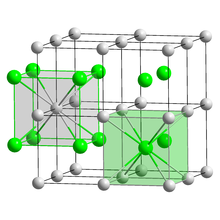
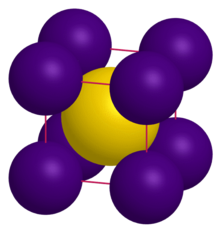
| Crystal structure | CsCl |
| Lattice constant | a = 4.24 Å |
| Except where otherwise noted, data are given for materials in their standard state (at 25 °C , 100 kPa).
| |
Caesium auride is the inorganic compound with the formula CsAu. It is the Cs salt of the unusual Au anion.
Preparation and reactions
CsAu is obtained by heating a stoichiometric mixture of caesium and gold. The two metallic-yellow liquids react to give a transparent yellow product. Despite being a compound of two metals, CsAu lacks metallic properties since it is a salt with localized charges; it instead behaves as a semiconductor with band gap 2.6 eV.
The compound hydrolyzes readily, yielding caesium hydroxide, metallic gold, and hydrogen.
- 2 CsAu + 2 H2O → 2 CsOH + 2 Au + H2
The solution in liquid ammonia is brown, and the ammonia adduct CsAu·NH3 is blue; the latter has ammonia molecules intercalated between layers of the CsAu crystal parallel to the (110) plane. Solutions undergo metathesis with tetramethylammonium loaded ion exchange resin to give tetramethylammonium auride.
References
- ^ Kienast, Gerhard; Verma, Jitendra; Klemm, Wilhelm (June 1961). "Das Verhalten der Alkalimetalle zu Kupfer, Silber und Gold". Zeitschrift für anorganische und allgemeine Chemie (in German). 310 (3): 143–169. doi:10.1002/zaac.19613100304.
- Peer, William J.; Lagowski, J. J. (1978). "Metal-Ammonia Solutions. 11. Au, a Solvated Transition Metal Anion". J. Am. Chem. Soc. 100: 6260–6261. doi:10.1021/ja00487a064.
- ^ Jansen, Martin (2005-11-30). "Effects of relativistic motion of electrons on the chemistry of gold and platinum". Solid State Sciences. 7 (12): 1464–1474. Bibcode:2005SSSci...7.1464J. doi:10.1016/j.solidstatesciences.2005.06.015.
- Norrby, Lars J. (February 1991). "Why is mercury liquid? Or, why do relativistic effects not get into chemistry textbooks?". Journal of Chemical Education. 68 (2): 110. Bibcode:1991JChEd..68..110N. doi:10.1021/ED068P110.
Further reading
- Jansen, Martin (2008). "The chemistry of gold as an anion". Chemical Society Reviews. 37 (9): 1826–1835. doi:10.1039/B708844M. PMID 18762832.—includes photograph of the compound.
| Gold compounds | |||
|---|---|---|---|
| Gold(-I) | |||
| Gold(I) |
| ||
| Gold(II) | |||
| Gold(I,III) | |||
| Gold(III) |
| ||
| Gold(V) | |||
| Gold(VI) |
| ||
| Caesium compounds | |
|---|---|