| |
| Names | |
|---|---|
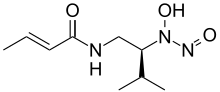
| Preferred IUPAC name (2E)-N-{(2S)-2--3-methylbutyl}but-2-enamide | |
| Identifiers | |
| CAS Number | |
| 3D model (JSmol) | |
| ChemSpider | |
| PubChem CID | |
| UNII | |
| CompTox Dashboard (EPA) | |
InChI
| |
SMILES
| |
| Properties | |
| Chemical formula | C9H17N3O3 |
| Molar mass | 215.253 g·mol |
| Melting point | 116 to 119 °C (241 to 246 °F; 389 to 392 K) |
| Acidity (pKa) | 5.1 |
| Except where otherwise noted, data are given for materials in their standard state (at 25 °C , 100 kPa). Infobox references | |
Dopastin is a chemical compound produced by the bacteria Pseudomonas No. BAC-125. It was first isolated and characterized in 1972. It is an inhibitor of the enzyme dopamine β-hydroxylase.
Dopastin can be prepared synthetically from L-valinol.
References
- ^ Merck Index, 11th Edition, 3417
- Iimura, H; Takeuchi, T; Kondo, S; Matsuzaki, M; Umezawa, H (1972). "Dopastin, an inhibitor of dopamine -hydroxylase". The Journal of Antibiotics. 25 (8): 497–500. doi:10.7164/antibiotics.25.497. PMID 4648494.
- H. Iinuma; M. Matsuzaki; T. Nagatsu; T. Takeuchi; H. Umezawa (1974). "Biochemical and biological studies on dopastin, an inhibitor of dopamine β-hydroxylase". Agric. Biol. Chem. 38 (11): 2107–2111. doi:10.1271/bbb1961.38.2107.
- Ohno, M.; Iinuma, H.; Yagisawa, N.; Shibahara, S.; Suhara, Y.; Kondo, S.; Maeda, K.; Umezawa, H. (1973). "Synthesis of dopastin, a dopamine ?-hydroxylase inhibitor of microbial origin". Journal of the Chemical Society, Chemical Communications (4): 147. doi:10.1039/C39730000147.