| |||
| Names | |||
|---|---|---|---|
| IUPAC name Fluoromethane | |||
| Other names
Freon 41 Methyl fluoride | |||
| Identifiers | |||
| CAS Number | |||
| 3D model (JSmol) | |||
| Abbreviations | R41 | ||
| Beilstein Reference | 1730725 | ||
| ChEBI | |||
| ChEMBL | |||
| ChemSpider | |||
| ECHA InfoCard | 100.008.907 | ||
| EC Number |
| ||
| Gmelin Reference | 391 | ||
| KEGG | |||
| MeSH | Fluoromethane | ||
| PubChem CID | |||
| UNII | |||
| UN number | UN 2454 | ||
| CompTox Dashboard (EPA) | |||
InChI
| |||
SMILES
| |||
| Properties | |||
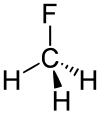
| Chemical formula | CH3F | ||
| Molar mass | 34.03 g/mol | ||
| Appearance | Colourless gas | ||
| Odor | pleasant, ether-like odour at high concentrations | ||
| Density | 1.4397 g/L 0.557 g/cm (liquid) at saturation pressure at 25 °C | ||
| Melting point | −137.8 °C (−216.0 °F; 135.3 K) | ||
| Boiling point | −78.4 °C (−109.1 °F; 194.8 K) | ||
| Solubility in water | 1.66 L/kg (2.295 g/L) | ||
| Vapor pressure | 3.3 MPa | ||
| Hazards | |||
| GHS labelling: | |||
| Pictograms | 
| ||
| Signal word | Danger | ||
| Hazard statements | H220 | ||
| Precautionary statements | P210, P377, P381, P403, P410+P403 | ||
| NFPA 704 (fire diamond) |
 | ||
| Except where otherwise noted, data are given for materials in their standard state (at 25 °C , 100 kPa).
| |||
Fluoromethane, also known as methyl fluoride, Freon 41, Halocarbon-41 and HFC-41, is a non-toxic, liquefiable, and flammable gas at standard temperature and pressure. It is made of carbon, hydrogen, and fluorine. The name stems from the fact that it is methane (CH4) with a fluorine atom substituted for one of the hydrogen atoms. It is used in semiconductor manufacturing processes as an etching gas in plasma etch reactors.
Fluoromethane (originally called "fluorohydrate of methylene") became the first organofluorine compound to be discovered when it was synthesized by French chemists Jean-Baptiste Dumas and Eugène-Melchior Péligot in 1835 by distilling dimethyl sulfate with potassium fluoride.
Composition
The compound is the lowest mass member of the hydrofluorocarbon (HFC) family, compounds which contain only hydrogen, fluorine, and carbon. These compounds are related to the chlorofluorocarbons (CFC), but since they do not contain chlorine, are not destructive to the ozone layer. Fluorocarbons are, however, potent greenhouse gasses, and the Kigali Amendment to the Montreal Protocol is an attempt to phase them out due to their contribution to global warming.
The C−F bond energy is 552 kJ/mol and its length is 0.139 nm. Its molecular geometry is tetrahedral. Its Dipole Moment is 1.85 D.
Its specific heat capacity (Cp) is 38.171 J·mol·K at 25 °C. The critical point of fluoromethane is at 44.9 °C (318.1 K) and 6.280 MPa.
See also
References
- ^ Record in the GESTIS Substance Database of the Institute for Occupational Safety and Health
- Siegemund, Günter; Schwertfeger, Werner; Feiring, Andrew; Smart, Bruce; Behr, Fred; Vogel, Herward; McKusick, Blaine (2002). "Fluorine Compounds, Organic". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi:10.1002/14356007.a11_349. ISBN 978-3527306732.
- Okazoe, Takashi (2009). "Overview on the history of organofluorine chemistry from the viewpoint of material industry". Proceedings of the Japan Academy, Series B. 85 (8): 276–289. Bibcode:2009PJAB...85..276O. doi:10.2183/pjab.85.276. PMC 3621566. PMID 19838009.
- Crochard (París); Arago, François; Gay-Lussac, Joseph Louis (1835). Annales de chimie et de physique (in French). Chez Crochard. p. 36.
- Fluoromethane CH3F
- "Explainer: hydrofluorocarbons saved the ozone layer, so why are we banning them?". 2 November 2017.
External links
| Halomethanes | |
|---|---|
| Unsubstituted | |
| Monosubstituted | |
| Disubstituted | |
| Trisubstituted | |
| Tetrasubstituted | |
| * Chiral compound. | |