| |
| Identifiers | |
|---|---|
| CAS Number | |
| 3D model (JSmol) | |
| ChemSpider | |
| PubChem CID |
|
| CompTox Dashboard (EPA) | |
InChI
| |
SMILES
| |
| Properties | |
| Chemical formula | WOF4 |
| Molar mass | 275.83 g/mol |
| Appearance | colourless crystals |
| Density | 5.07 g/cm |
| Melting point | 110 °C (230 °F; 383 K) |
| Boiling point | 185 °C (365 °F; 458 K) |
| Solubility in water | reacts |
| Solubility | soluble in chloroform sparingly soluble in carbon disulfide |
| Structure | |
| Crystal structure | monoclinic |
| Related compounds | |
| Other anions | Tungsten(VI) oxytetrachloride Tungsten(VI) oxytetrabromide |
| Except where otherwise noted, data are given for materials in their standard state (at 25 °C , 100 kPa).
| |
Tungsten oxytetrafluoride is an inorganic compound with the formula WOF4. It is a colorless diamagnetic solid. The compound is one of many oxides of tungsten. It is usually encountered as product of the partial hydrolysis of tungsten hexafluoride.
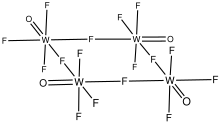
Structure
As confirmed by X-ray crystallography, WOF4 crystallizes as a tetramer. The oxides are terminal, and four of the fluorides are bridging. Its structure is similar to those for niobium pentafluoride and tantalum pentafluoride. In contrast, molybdenum oxytetrafluoride adopts a polymeric structure, although again the fluorides bridge and the oxides are terminal.
In the gas state, this molecule is a monomer. It can form complexes with acetonitrile and other compounds.
Preparation
Tungsten(VI) oxytetrafluoride can be synthesized by the reaction of fluorine and tungsten trioxide.
It can also be obtained by treating tungsten with a mixture of oxygen and fluorine at high temperatures. Partial hydrolysis of tungsten hexafluoride will also produce WOF4.
- WF6 + H2O → WOF4 + 2 HF
The reaction of tungsten(VI) oxytetrachloride and hydrogen fluoride will also produce WOF4.
- WOCl4 + 4HF → WOF4 + 4HCl
WOF4 can also prepared by the reaction of lead(II) fluoride and tungsten trioxide at 700 °C.
- 2PbF2 + WO3 → WOF4 + 2PbO
Tungsten(VI) oxytetrafluoride hydrolyzes into tungstic acid.
- WOF4 + 2 H2O → WO3 + 4 HF
References
- ^ Perry, Dale L.; Phillips, Sidney L. (1995). Handbook of inorganic compounds. Boca Raton: CRC Press. p. 428. ISBN 0-8493-8671-3. OCLC 32347397.
- ^ Haynes, William M.; Lide, David R.; Bruno, Thomas J. (2017). CRC handbook of chemistry and physics : a ready-reference book of chemical and physical data. Boca Raton, Florida. p. 104. ISBN 978-1-4987-5429-3. OCLC 957751024.
{{cite book}}: CS1 maint: location missing publisher (link) - ^ Lassner, Erik; Schubert, Wolf-Dieter (1999). Tungsten : Properties, Chemistry, Technology of the Element, Alloys, and Chemical Compounds. Boston, MA. p. 168. ISBN 1-4615-4907-8. OCLC 1113605323.
{{cite book}}: CS1 maint: location missing publisher (link) - ^ Turnbull, Douglas; Chaudhary, Praveen; Leenstra, Dakota; Hazendonk, Paul; Wetmore, Stacey D.; Gerken, Michael (2020). "Reactions of Molybdenum and Tungsten Oxide Tetrafluoride with Sulfur(IV) Lewis Bases: Structure and Bonding in [WOF4]4, MOF4(OSO), and [SF3][M2O2F9] (M = Mo, W)". Inorganic Chemistry. 59 (23): 17544–17554. doi:10.1021/acs.inorgchem.0c02783. PMID 33200611. S2CID 226989898.
- Edwards, A. J.; Steventon, B. R. (1968). "Fluoride crystal structures. Part II. Molybdenum oxide tetrafluoride". Journal of the Chemical Society A: Inorganic, Physical, Theoretical: 2503. doi:10.1039/j19680002503.
- Johnson, B. F. G. (1976). Inorganic chemistry of the transition elements. Volume 4, A review of the literature published between October 1973 and September 1974. London: Chemical Society. p. 138. ISBN 978-1-84755-645-5. OCLC 820579758.
- Levason, William; Reid, Gillian; Zhang, Wenjian (2016). "Coordination complexes of the tungsten(VI) oxide fluorides WOF4 and WO2F2 with neutral oxygen- and nitrogen-donor ligands". Journal of Fluorine Chemistry. 184. Elsevier BV: 50–57. doi:10.1016/j.jfluchem.2016.02.003. ISSN 0022-1139.
- Arnaudet, Lucile; Bougon, Roland; Charpin, Pierrette; Isabey, Jacques; Lance, Monique; Nierlich, Martine; Vigner, Julien (1989). "Preparation, characterization, and crystal structure of the adducts WOF4.nC5H5N (n = 1, 2)". Inorganic Chemistry. 28 (2). American Chemical Society (ACS): 257–262. doi:10.1021/ic00301a020. ISSN 0020-1669.
- ^ Mendicino, L.; Electrochemical Society. Dielectric Science and Technology Division; Electrochemical Society. Meeting; Symposium on Environmental Issues with Materials and Processes in the Electronics and Semiconductor Industries (2001). Environmental issues with materials and processes for the electronics and semiconductor industries : proceedings of the fourth international symposium. Pennington, NJ: Electrochemical Society. p. 180. ISBN 1-56677-312-1. OCLC 48710248.
| Tungsten compounds | |||||
|---|---|---|---|---|---|
| Tungsten(0) | |||||
| Tungsten(II) | |||||
| Tungsten(III) | |||||
| Tungsten(IV) | |||||
| Tungsten(V) | |||||
| Tungsten(VI) |
| ||||