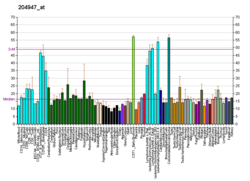
Protein-coding gene in the species Homo sapiens
Transcription factor E2F1 is a protein that in humans is encoded by the E2F1 gene .
Function
The protein encoded by this gene is a member of the E2F family of transcription factors . The E2F family plays a crucial role in the control of cell cycle and action of tumor suppressor proteins and is also a target of the transforming proteins of small DNA tumor viruses. The E2F proteins contain several evolutionarily conserved domains found in most members of the family. These domains include a DNA binding domain , a dimerization domain which determines interaction with the differentiation regulated transcription factor proteins (DP), a transactivation domain enriched in acidic amino acids, and a tumor suppressor protein association domain which is embedded within the transactivation domain. This protein and another 2 members, E2F2 and E2F3, have an additional cyclin binding domain. This protein binds preferentially to retinoblastoma protein pRB in a cell-cycle dependent manner. It can mediate both cell proliferation and p53 -dependent/independent apoptosis .
Transcription
E2F1 promoter => E2F1
Interactions
E2F1 has been shown to interact with:
ARID3A ,CUL1 ,Cyclin A1 ,Cyclin A2 ,GTF2H1 ,MDM4 ,NCOA6 ,NDN ,NPDC1 ,PURA ,PHB ,RB1 ,UDG ,RBL1 ,SKP2 ,SP1 ,SP2 ,SP3 ,SP4 ,TFDP1 TOPBP1 ,TP53BP1 , andUBC .See also
References
^ GRCh38: Ensembl release 89: ENSG00000101412 – Ensembl , May 2017
^ GRCm38: Ensembl release 89: ENSMUSG00000027490 – Ensembl , May 2017
"Human PubMed Reference:" . National Center for Biotechnology Information, U.S. National Library of Medicine ."Mouse PubMed Reference:" . National Center for Biotechnology Information, U.S. National Library of Medicine .Neuman E, Sellers WR, McNeil JA, Lawrence JB, Kaelin WG (December 1996). "Structure and partial genomic sequence of the human E2F1 gene". Gene . 173 (2): 163–9. doi :10.1016/0378-1119(96)00184-9 . PMID 8964493 .
"Entrez Gene: E2F1 E2F transcription factor 1" .Li CG, Nyman JE, Braithwaite AW, Eccles MR (December 2011). "PAX8 promotes tumor cell growth by transcriptionally regulating E2F1 and stabilizing RB protein" . Oncogene . 30 (48): 4824–34. doi :10.1038/onc.2011.190 . PMC 3229668 . PMID 21602887 .
^ Suzuki M, Okuyama S, Okamoto S, Shirasuna K, Nakajima T, Hachiya T, Nojima H, Sekiya S, Oda K (August 1998). "A novel E2F binding protein with Myc-type HLH motif stimulates E2F-dependent transcription by forming a heterodimer" . Oncogene . 17 (7): 853–65. doi :10.1038/sj.onc.1202163 . PMID 9780002 .
^ Marti A, Wirbelauer C, Scheffner M, Krek W (May 1999). "Interaction between ubiquitin-protein ligase SCFSKP2 and E2F-1 underlies the regulation of E2F-1 degradation". Nat. Cell Biol . 1 (1): 14–9. doi :10.1038/8984 . PMID 10559858 . S2CID 8884226 .
Yang R, Müller C, Huynh V, Fung YK, Yee AS, Koeffler HP (March 1999). "Functions of cyclin A1 in the cell cycle and its interactions with transcription factor E2F-1 and the Rb family of proteins" . Mol. Cell. Biol . 19 (3): 2400–7. doi :10.1128/mcb.19.3.2400 . PMC 84032 . PMID 10022926 .
Xu M, Sheppard KA, Peng CY, Yee AS, Piwnica-Worms H (December 1994). "Cyclin A/CDK2 binds directly to E2F-1 and inhibits the DNA-binding activity of E2F-1/DP-1 by phosphorylation" . Mol. Cell. Biol . 14 (12): 8420–31. doi :10.1128/MCB.14.12.8420 . PMC 359381 . PMID 7969176 .
Vandel L, Kouzarides T (August 1999). "Residues phosphorylated by TFIIH are required for E2F-1 degradation during S-phase" . EMBO J . 18 (15): 4280–91. doi :10.1093/emboj/18.15.4280 . PMC 1171504 . PMID 10428966 .
Strachan GD, Jordan-Sciutto KL, Rallapalli R, Tuan RS, Hall DJ (February 2003). "The E2F-1 transcription factor is negatively regulated by its interaction with the MDMX protein" . J. Cell. Biochem . 88 (3): 557–68. doi :10.1002/jcb.10318 . PMID 12532331 . S2CID 38805122 .
Kong HJ, Yu HJ, Hong S, Park MJ, Choi YH, An WG, Lee JW, Cheong J (November 2003). "Interaction and functional cooperation of the cancer-amplified transcriptional coactivator activating signal cointegrator-2 and E2F-1 in cell proliferation". Mol. Cancer Res . 1 (13): 948–58. PMID 14638867 .
^ Taniura H, Taniguchi N, Hara M, Yoshikawa K (January 1998). "Necdin, a postmitotic neuron-specific growth suppressor, interacts with viral transforming proteins and cellular transcription factor E2F1" . J. Biol. Chem . 273 (2): 720–8. doi :10.1074/jbc.273.2.720 . PMID 9422723 .
Kuwako K, Taniura H, Yoshikawa K (January 2004). "Necdin-related MAGE proteins differentially interact with the E2F1 transcription factor and the p75 neurotrophin receptor" . J. Biol. Chem . 279 (3): 1703–12. doi :10.1074/jbc.M308454200 . PMID 14593116 .
Sansal I, Dupont E, Toru D, Evrard C, Rouget P (October 2000). "NPDC-1, a regulator of neural cell proliferation and differentiation, interacts with E2F-1, reduces its binding to DNA and modulates its transcriptional activity" . Oncogene . 19 (43): 5000–9. doi :10.1038/sj.onc.1203843 . PMID 11042687 .
Darbinian N, Gallia GL, Kundu M, Shcherbik N, Tretiakova A, Giordano A, Khalili K (November 1999). "Association of Pur alpha and E2F-1 suppresses transcriptional activity of E2F-1" . Oncogene . 18 (46): 6398–402. doi :10.1038/sj.onc.1203011 . PMID 10597240 .
Joshi B, Ko D, Ordonez-Ercan D, Chellappan SP (December 2003). "A putative coiled-coil domain of prohibitin is sufficient to repress E2F1-mediated transcription and induce apoptosis". Biochem. Biophys. Res. Commun . 312 (2): 459–66. doi :10.1016/j.bbrc.2003.10.148 . PMID 14637159 .
Fusaro G, Dasgupta P, Rastogi S, Joshi B, Chellappan S (November 2003). "Prohibitin induces the transcriptional activity of p53 and is exported from the nucleus upon apoptotic signaling" . J. Biol. Chem . 278 (48): 47853–61. doi :10.1074/jbc.M305171200 . PMID 14500729 .
Wang S, Zhang B, Faller DV (June 2002). "Prohibitin requires Brg-1 and Brm for the repression of E2F and cell growth" . EMBO J . 21 (12): 3019–28. doi :10.1093/emboj/cdf302 . PMC 126057 . PMID 12065415 .
Wang S, Nath N, Fusaro G, Chellappan S (November 1999). "Rb and prohibitin target distinct regions of E2F1 for repression and respond to different upstream signals" . Mol. Cell. Biol . 19 (11): 7447–60. doi :10.1128/mcb.19.11.7447 . PMC 84738 . PMID 10523633 .
^ Dyson N, Dembski M, Fattaey A, Ngwu C, Ewen M, Helin K (December 1993). "Analysis of p107-associated proteins: p107 associates with a form of E2F that differs from pRB-associated E2F-1" . J. Virol . 67 (12): 7641–7. doi :10.1128/JVI.67.12.7641-7647.1993 . PMC 238233 . PMID 8230483 .
Nicolas E, Ait-Si-Ali S, Trouche D (August 2001). "The histone deacetylase HDAC3 targets RbAp48 to the retinoblastoma protein" . Nucleic Acids Res . 29 (15): 3131–6. doi :10.1093/nar/29.15.3131 . PMC 55834 . PMID 11470869 .
Pardo PS, Leung JK, Lucchesi JC, Pereira-Smith OM (December 2002). "MRG15, a novel chromodomain protein, is present in two distinct multiprotein complexes involved in transcriptional activation" . J. Biol. Chem . 277 (52): 50860–6. doi :10.1074/jbc.M203839200 . PMID 12397079 .
^ Choubey D, Li SJ, Datta B, Gutterman JU, Lengyel P (October 1996). "Inhibition of E2F-mediated transcription by p202" . EMBO J . 15 (20): 5668–78. doi :10.1002/j.1460-2075.1996.tb00951.x . PMC 452311 . PMID 8896460 .
Fajas L, Paul C, Zugasti O, Le Cam L, Polanowska J, Fabbrizio E, Medema R, Vignais ML, Sardet C (July 2000). "pRB binds to and modulates the transrepressing activity of the E1A-regulated transcription factor p120E4F" . Proc. Natl. Acad. Sci. U.S.A . 97 (14): 7738–43. Bibcode :2000PNAS...97.7738F . doi :10.1073/pnas.130198397 . PMC 16614 . PMID 10869426 .
^ Wu CL, Zukerberg LR, Ngwu C, Harlow E, Lees JA (May 1995). "In vivo association of E2F and DP family proteins" . Mol. Cell. Biol . 15 (5): 2536–46. doi :10.1128/mcb.15.5.2536 . PMC 230484 . PMID 7739537 .
Walsh M, Shue G, Spidoni K, Kapoor A (March 1995). "E2F-1 and a cyclin-like DNA repair enzyme, uracil-DNA glycosylase, provide evidence for an auto-regulatory mechanisms for transcription" . J. Biol. Chem . 270 (10): 5289–98. doi :10.1074/jbc.270.10.5289 . PMID 7534293 .
^ Rotheneder H, Geymayer S, Haidweger E (November 1999). "Transcription factors of the Sp1 family: interaction with E2F and regulation of the murine thymidine kinase promoter". J. Mol. Biol . 293 (5): 1005–15. doi :10.1006/jmbi.1999.3213 . PMID 10547281 .
Lin SY, Black AR, Kostic D, Pajovic S, Hoover CN, Azizkhan JC (April 1996). "Cell cycle-regulated association of E2F1 and Sp1 is related to their functional interaction" . Mol. Cell. Biol . 16 (4): 1668–75. doi :10.1128/MCB.16.4.1668 . PMC 231153 . PMID 8657142 .
Karlseder J , Rotheneder H, Wintersberger E (April 1996). "Interaction of Sp1 with the growth- and cell cycle-regulated transcription factor E2F" . Mol. Cell. Biol . 16 (4): 1659–67. doi :10.1128/mcb.16.4.1659 . PMC 231152 . PMID 8657141 .Sardet C, Vidal M, Cobrinik D, Geng Y, Onufryk C, Chen A, Weinberg RA (March 1995). "E2F-4 and E2F-5, two members of the E2F family, are expressed in the early phases of the cell cycle" . Proc. Natl. Acad. Sci. U.S.A . 92 (6): 2403–7. Bibcode :1995PNAS...92.2403S . doi :10.1073/pnas.92.6.2403 . PMC 42492 . PMID 7892279 .
Helin K, Wu CL, Fattaey AR, Lees JA, Dynlacht BD, Ngwu C, Harlow E (October 1993). "Heterodimerization of the transcription factors E2F-1 and DP-1 leads to cooperative trans-activation" . Genes Dev . 7 (10): 1850–61. doi :10.1101/gad.7.10.1850 . PMID 8405995 .
Liu K, Lin FT, Ruppert JM, Lin WC (May 2003). "Regulation of E2F1 by BRCT domain-containing protein TopBP1" . Mol. Cell. Biol . 23 (9): 3287–304. doi :10.1128/mcb.23.9.3287-3304.2003 . PMC 153207 . PMID 12697828 .
Yu X, Chini CC, He M, Mer G, Chen J (October 2003). "The BRCT domain is a phospho-protein binding domain". Science . 302 (5645): 639–42. Bibcode :2003Sci...302..639Y . doi :10.1126/science.1088753 . PMID 14576433 . S2CID 29407635 .
Zhou F, Zhang L, Wang A, Song B, Gong K, Zhang L, Hu M, Zhang X, Zhao N, Gong Y (May 2008). "The association of GSK3 beta with E2F1 facilitates nerve growth factor-induced neural cell differentiation" . J. Biol. Chem . 283 (21): 14506–15. doi :10.1074/jbc.M706136200 . PMID 18367454 .
Further reading
Dupont E, Sansal I, Toru D, Evrard C, Rouget P (1997). "". C. R. Séances Soc. Biol. Fil . 191 (1): 95–104. PMID 9181131 . Stevens C, La Thangue NB (2005). "The emerging role of E2F-1 in the DNA damage response and checkpoint control" . DNA Repair (Amst.) . 3 (8–9): 1071–9. doi :10.1016/j.dnarep.2004.03.034 . PMID 15279795 . Zhang Z, Wang H, Li M, Rayburn E, Agrawal S, Zhang R (2006). "Novel MDM2 p53-independent functions identified through RNA silencing technologies". Ann. N. Y. Acad. Sci . 1058 (1): 205–14. Bibcode :2005NYASA1058..205Z . doi :10.1196/annals.1359.030 . PMID 16394138 . S2CID 35683657 . Schild C, Wirth M, Reichert M, Schmid RM, Saur D, Schneider G (July 2009). "PI3K signaling maintains c-myc expression to regulate transcription of E2F1 in pancreatic cancer cells". Mol. Carcinog . 48 (12): 1149–58. doi :10.1002/mc.20569 . PMID 19603422 . S2CID 41545085 . External links
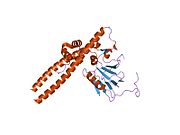
PDB gallery
2aze : Structure of the Rb C-terminal domain bound to an E2F1-DP1 heterodimer
This article incorporates text from the United States National Library of Medicine , which is in the public domain .
Categories :
Text is available under the Creative Commons Attribution-ShareAlike License. Additional terms may apply.
**DISCLAIMER** We are not affiliated with Wikipedia, and Cloudflare.
The information presented on this site is for general informational purposes only and does not constitute medical advice.
You should always have a personal consultation with a healthcare professional before making changes to your diet, medication, or exercise routine.
AI helps with the correspondence in our chat.
We participate in an affiliate program. If you buy something through a link, we may earn a commission 💕
↑